Abstract
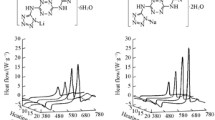
The compounds [(LR2Sn)2O] {R=CH3 (Me), n-C4H9(Bn); L=C6H5CH(OH)COO−}were studied by thermogravimetric and differential scanning calorimetry in a dynamic atmosphere of helium. The thermal decomposition mechanisms are similar for both compounds and occur in two consecutive steps. The TG curves of the complexes suggest the liberation of the ligand L in the first step, with probable formation of a tin oxide R2SnO intermediate. At the end of the second step free tin is obtained in accordance with the stoichiometry of the related compounds. An inverse relation betweenΔH fusion and solubility of the compounds suggests a polymeric structure for the compound with Bn ligand in relation to the compound with M ligand.
Similar content being viewed by others
References
T. P. Lockart, W. F. Manders and E. M. Holt, J. Am. Chem. Soc., 108 (1986) 6611.
V. B. Mokal, V. K. Jain and E. R. T. Tiekink, J. Organomet. Chem., 407 (1991) 173.
C. S. Parulekar, V. K. Jain, T. K. Das and A. R. Gupta, J. Organomet. Chem., 372 (1989) 372.
G. K. Sandhu, S. P. Verma and E. R. T. Tiekink, J. Organomet. Chem., 393 (1990) 195.
E. R. T. Tiekink, J. Organomet. Chem., 408 (1991) 323.
J. Holecek, M. Nadvornik, K. Handlir and A. Licka, J. Organomet. Chem., 315 (1986) 299.
M. A. Mesubi, Spectrochim. Acta A, 38 (1976) 989.
M. A. Mesubi and G. A. Olatunji, Termochim. Acta, 67 (1983) 307.
G. K. Sandhu and R. Hundal, J. Organomet. Chem., 430 (1992) 15.
A. G. Davies, Organotin Chemistry, VCH Verlagsgesellschaft 1997.
A. G. Davies and S. Karpel, Comprehensive Organometallic Chemistry, v.1, Pergamon Press 1982, p. 273.
I. Omae, Organotin Chemistry, Elsevier 1989, p. 335-337.
A. K. Carlos Dias, Dissertation-Master Program in Agrochemistry and Agrobiochemistry, UFLA (Brazil) 1999.
I. Haidua and C. Silvestu, Coord. Chem. Ver., 99 (1990) 253.
M. A. Mesubi, Spectrochim. Acta, 67 (1982) 989.
V. R. Terra, R. S. Barbiéri, P. A. Castelo Branco and A. Abras, Ecl. Chim., 23 (1998) 17.
N. W. G. Debye, D. E. Fenton, S. E. Ulrich and J. Zuckerman, J. Organomet. Chem., 28 (1971) 339.
R. Cefalu, R. Bosco, R. Bonati, F. Maggio and R. Barbieri, Z. Anorg. Allg. Chem., 376 (1970) 180.
E. V. Van Der Berghe, G. P. Van der Kelen and J. Albrecht, Inorg. Chim. Acta, 2 (1968) 89.
R. V. Parish, Prog. Inorg. Chem., 15 (1992) 101.
G. K. Sandhu, R. Gupta, S. S. Sandhu, L. S. Moore and R. V. Parish, J. Organometal. Chem., 311 (1986) 281.
T. Yano, K. Nakashima, J. Otera and R. Okawara, Organometallics, 4 (1985) 1501.
D. C. Gross, Inorg. Chem., 28 (1989) 2355.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Terra, V.R., Barbiéri, R.S., Dias, A.K.C. et al. Thermal Analyses of Diorganotin dl-Mandelates. Journal of Thermal Analysis and Calorimetry 67, 453–458 (2002). https://doi.org/10.1023/A:1013988930942
Issue Date:
DOI: https://doi.org/10.1023/A:1013988930942