Abstract
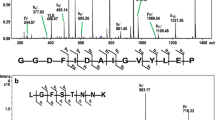
In this work, we characterized chemically the N-acetyl-D-galactosamine specific lectin from Amaranthus leucocarpus syn hypocondriacus lectin (ALL). It is a dimeric glycoprotein composed by three isoforms with pl at 4.8, 4.9, and 5.2. Circular dichroism analysis indicated that the secondary structure of ALL contains 45% of β-sheet and 5% of α-helix. Amino acid sequence of the purified lectin and its isoforms was determined from peptides obtained after trypsin digestion by MALDI-TOF (Matrix assisted laser desorption ionization-time of flight). The tryptic peptides prepared from the purified lectin and the three isoforms showed different degrees (80 to 83%) of identity with the amino acid sequence belonging to a previously described high nutritional value protein from A. hypocondriacus not shown at the time to be a lectin. Furthermore, analysis of tryptic peptides obtained from ALL previously treated with peptide N-glycosidase, revealed a 93% identity with the aforementioned protein. Presence of N-glycosidically linked glycans of the oligomannosidic type and, in minor proportion, of the N-acetyllactosaminic type glycans was determined by affinity chromatography on immobilized Con A.
Similar content being viewed by others
References
Sharon N, Lis H, Legume lectins, a large family of homologous proteins, FASEB J 4, 3198–208 (1990).
Weiss I, Drickramer K, Structural basis of the lectin-carbohydrate recognition, Annu Rev Biochem 65, 441–73 (1996).
Van Damme EJ, Peumans WJ, Barre A, Rouge P, Plant lectins: A composite of several distinct families of structurally and evolutionary related proteins with diverse biological roles, Crit Rev Biochem Mol Biol 33, 209–58 (1998).
Rinderle SJ, Goldstein IJ, Matta KL, Ratcliffe RM, Isolation and characterization of amaranthin, a lectin present in the seeds of Amaranthus caudatus, that recognizes the T-(or cryptic T)-antigen, J Biol Chem 264, 16123–31 (1989).
Calderon de la Barca A, Vazquez-Moreno L, Amaranthus cruentus lectin: Purification, stability and some biochemical properties, J Food Biochem 12, 117–26 (1988).
Zenteno E, Ochoa JL, Isolation and characterization of Amaranthus leucocarpus lectin, Phytochemistry 27, 313–7 (1988).
Zenteno E, Lascurain R, Montaño LF, Vazquez L, Debray H, Montreuil J, Specificity of Amaranthus leucocarpus lectin, Glycoconjugate J 9, 204–8 (1992).
Zenteno E, Ochoa JL, Parra C, Montaño LF, Rayon I, Maldonado G, Ruiz B, Carvajal R, Mitogenic immunosuppressive and phagocytic activity of Machaerocerus eruca and Amaranthus leucocarpus lectin. In Lectins-biology, Biochemistry, Clinical Biochemistry, Vol. 4, edited by Boög-Hansen TC, Breborowicz J, (Walter de Gruyter, 1985), pp. 537–46.
Boland CR, Chen YF, Rinderle SJ, Resau JH, Luk GD, Lynch HT, Goldstein IJ, Use of the lectin from Amaranthus caudatus as a histochemical probe of proliferating colonic epithelial cells, Cancer Res 51, 657–65 (1991).
Lascurain R, Chávez R, Gorocica P, Pérez A, Montaño LF, Zenteno E, Recognition of CD4+ mouse medullary thymocyte subpopulation by Amaranthus leucocarpus lectin, Immunology 83, 410–3 (1994).
Lascurain R, Porras F, Baáz R, Chávez R, Martínez S, Álvarez G, Zenteno E, Amaranthus leucocarpus lectin recognizes human naive T cell subpopulations, Immunol Invest 26, 579–87 (1997).
Guevara J, Espinosa B, Zenteno E, Vázquez L, Luna J, Perry G, Mena R, Altered glycosylation pattern of proteins in Alzheimer disease, J Neuropath Exp Neurol 57, 905–14 (1998).
Hernández P, Bacilio M, Porras F, Juárez S, Debray H, Zenteno E, Ortiz B, A comparative study on the purification of the Amaranthus leucocarpus syn. hypocondriacus lectin, Prep Biochem Biotechnol 29, 219–34 (1999).
Kuster B, Wheler SF, Hunter AP, Dweek RA, Harvey DJ, Sequencing of N-linked oligosaccharides directly from protein gels: in-gel deglycosylation followed by matrix-assisted laser desorption/ionization mass spectrometry and normal-phase highperformance liquid chromatography, Anal Biochem 250, 82–101 (1977).
Bradford MM, A rapid and sensitive method for quantitation of microgram quantities of proteins utilizing the principle of protein dye-binding, Anal Biochem 72, 248–54 (1976).
Zanetta JP, Timmerman P, Leroy Y, Gas–liquid chromatography of the heptafluorobutirate derivatives of O-methyl-glycosides on capillary columns: a method for the quantitative determination of monosaccharide composition of glycoproteins and glycolipids, Glycobiology 9, 255–66 (1999).
Catsimpoolas N, Micro isoelectric focusing in polyacrylamide gel columns, Anal Biochem 26, 480–4 (1968).
Laemmli UK, Favra M, Maturation of the head of bacteriophage T4. I. DNA packaging events, J Mol Biol 80, 575–99 (1973).
Hellman U, Wernstedt C, Gonez J, Heldin CH, Improvement of an “In-Gel” digestion procedure for the micropreparation of internal protein fragments for amino acid sequencing, Anal Biochem 224, 451–5 (1995).
Geourjon C, Deleage G, SOPMA: significant improvements in protein secondary structure prediction by consensus prediction from multiple alignments, Comput Appl Biosci 11, 681–4 (1995).
Garnier J, Gibrat JF, Robson B, GOR secondary structure prediction method version IV. In Methods in Enzymology, Vol. 266, edited by Doolittle RF, (1996), pp. 540–53.
Jones DT, Protein secondary structure prediction based on positionspecific scoring matrices, J Mol Biol 292, 195–202 (1999).
Transue TR, Smith AK, Mo H, Goldstein IJ, Saper, MA, Structure of benzyl T-antigen disaccharide bound to Amaranthus caudatus agglutinin, Nat Struct Biol 4, 779–83 (1997).
Raina A, Datta A, Molecular cloning of a gene encoding a seedspecific protein with nutritionally balanced amino acid composition from Amaranthus, Proc Natl Acad Sci USA 89, 11774–8 (1992).
Calderon de la Barca A, Ochoa JL, Valencia ME, Effect of the extraction of a hemagglutinin on the nutritive value of Amaranthus leucocarpus seeds, J Food Sci 50, 1700–8 (1985).
Calderon de la Barca A, Zenteno E, Ochoa JL, Valencia M, Carvajal R, The removal of Amaranthus leucocarpus lectin does not affect the nutritional value of the seed meal. In Lectins-Biology, Biochemistry, Clinical Biochemistry, Vol. 4, edited by Bög-Hansen TC, Breborowics J, (Walter de Gruyter, 1985), pp. 531–6.
Rini M, Lectin structure, Annu Rev Biomol Struc 24, 551–77 (1995).
Sharma V, Surolia A, Analysis of carbohydrate recognition by legume lectin: Size of the combining site loops and their primary specificity, J Mol Biol 267, 433–45 (1997).
Rinderle SJ, Goldstein IJ, Remsen EE, Physicochemical properties of Amaranthin, the lectin from Amaranthus caudatus seeds, Biochemistry 29, 10555–61 (1990).
Debray H, Decout D, Strecker G, Spik G, Montreuil J, Specificity of twelve lectins towards oligosaccharides and glycopeptides related to N-glycosylproteins, Eur J Biochem 117, 41–55 (1981).
Kuhn P, Guan C, Cui T, Tarentino AL, Plummer TH Jr, Van Roey P, Active site and oligosaccharide recognition residues of peptide-N-(N-acetyl-D-glucosaminyl)asparagine amidase F, J Biol Chem 270, 29493–7 (1995).
Perez Campos E, Sierra C, Lascurain R, Espinoza B, Bouquelet S, Debray H, Zenteno E, The erythroagglutinin from Phaseolus coccineus Var. Alubia: chemical characterization, sugar specificity and effect on coagulation factors, J Agric Food Chem 45, 3747–52 (1997).
Capon C, Piller F, Wieruszeski JM, Leroy I, Fournet B, The glycomannosidic structure from jacalin, Artocarpus integrifolia lectin, Carbohydr Res 199, 121–7 (1990).
Fournet B, Leroy Y, Wieruszeski JM, Montreuil J, Poretz RD, Goldberg R, Primary structure of an N-glycosidic carbohydrate unit derived from Sophora japonica lectin, Eur J Biochem 166, 321–4 (1987).
Ortega M, Sanchez C, Chacon E, Estrada R, Masso F, Montaño LF, Rendon J, Zenteno E, Purification and characterization of a lectin from Erythrina americana by affinity chromatography, Plant Science 72, 133–40 (1990).
Debray H, Wieruszeski JM, Strecker G, Franz H, Structural analysis of the carbohydrate chains isolated from mistletoe (Viscum album) lectin I, Carbohydr Res 236, 135–43 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hernández, P., Debray, H., Jaekel, H. et al. Chemical characterization of the lectin from Amaranthus leucocarpus syn. hypocondriacus by 2-D proteome analysis. Glycoconj J 18, 321–329 (2001). https://doi.org/10.1023/A:1013760915738
Issue Date:
DOI: https://doi.org/10.1023/A:1013760915738