Abstract
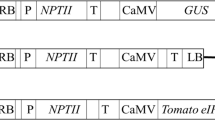
Potato mop-top virus (PMTV) RNA 2 contains a triple gene block (TGB) that consists of a set of three overlapping genes. These genes encode three proteins necessary for viral cell-to-cell movement. The gene encoding for the second triple gene block protein, TGB2, was mutated in a region that is highly conserved among TGB2 proteins from different viruses. The mutated TGB2 gene, under transcriptional control of the 35S promoter from cauliflower mosaic virus, was transformed into potato (Solanum tuberosum cv. Hulda) by Agrobacterium-mediated transformation. Ten of the transgenic lines obtained, all of which were shown to transcribe the introduced gene, were evaluated for resistance to PMTV in a field trial. Tubers from the field were analysed for virus content by ELISA. In seven of the transgenic lines analysed there was a reduction in ELISA values relative to a non-transgenic control line. In the three lines that had the lowest ELISA readings this reduction was manifested both as a reduction in the proportion of infected tubers and as a reduction in virus levels in infected tubers. In the line showing the highest level of resistance, the total ELISA value was reduced by 79%. These results demonstrate that the introduction of a mutated TGB2 gene into potato can confer increased resistance to PMTV.
Similar content being viewed by others
References
Anderson J.M., Palukaitis P. and Zaitlin M: A defective replicase gene induces resistance to cucumber mosaic virus in transgenic tobacco plants. Proc. Natl. Acad. Sci. USA 89: 8759–8763.
Barker H., Reavy B. and McGeachy K.D. 1998a. High level of resistance in potato to potato mop-top virus induced by transformation with the coat protein gene. Eur. J. Plant Path. 104: 737–740.
Barker H., Reavy B., McGeachy K.D. and Dawson S. 1998b. Transformation of Nicotiana benthamiana with the potato mop-top virus coat protein gene produces a novel resistance phenotype mediated by the coat protein. Mol. Plant-Microbe Interact. 11: 626–633.
Beachy R.N. 1997. Mechanism and applications of pathogenderived resistance in transgenic plants. Curr. Opin. Biotechnol. 8: 215–220.
Beck E., Ludwig G., Auerswald E.A., Reiss B. and Schaller H. 1982. Nucleotide sequence and exact localization of the neomycin phosphotransferase gene from transposon Tn5. Gene 19: 327–336.
Beck D.L., Guilford P.J., Voot D.M., Andersen M.T. and Forster R.L.S. 1991. Triple gene block proteins of white clover mosaic potexvirus are required for transport. Virology 183: 695–702.
Beck D.L., Van Dollewerd C.J., Lough T.J., Balmori E., Voot D.M., Andersen M.T., O'Brien I.E.W. and Forster R.L.S. 1994. Disruption of virus movement confers broad-spectrum resistance against systemic infection by plant viruses with a triple gene block. Proc. Natl. Acad. Sci. USA 91: 10310–10314.
Collier G.H., Wurr D.C.E. and Huntington V.C. 1978. The effect of calcium nutrition on the incidence of internal rust spot in potato. J. Agric. Sci. 91: 249–243.
Davies H.V. and Millard P. 1985. Fractionation and distribution of calcium in sprouting and non-sprouting potato tubers. Ann Bot. 56: 745–754.
Erhardt M., Herzog E., Lauber E., Fritsch C., Guilley H., Jonard G. and Bouzoubaa S. 1999. Transgenic plants expressing the TGB1 protein of peanut clump virus complement movement of TGB1-defective peanut clump virus but not of TGB1-defective beet necrotic yellow vein virus. Plant Cell Rep. 18: 614–619.
Gilmer D., Gafny R., Ding B., Wolf S., LucasW.J. and Beachy R.N. 1992. Efficient cell-to-cell movement of beet necrotic yellow vein virus requires 3′ proximal genes located on RNA2. Virology 189: 40–47.
Herzog E., Hemmer O., Hauser S., Meyer G., Bouzoubaa S. and Fritsch C. 1998. Identification of genes involved in replication and movement of peanut clump virus. Virology 248: 312–322.
Hofvander P., Persson P.T., Tallberg A. and Wikström O. 1991. Genetically engineered modification of potato to form amylopectin-type starch. PCT application, WO 92/11376.
Jones R.A.C. and Harrison B.D. 1969. The behaviour of potato mop-top virus in soil, and evidence for its transmission by Spongospora subterranea (Wallr.) Lagerh. Ann. Appl. Biol. 63: 1–17.
Lapidot M., Gafny R., Ding B., Wolf S., Lucas W.J. and Beachy R.N. 1993. A dysfunctional movement protein of tobacco mosaic virus that partially modifies the plasmodesmata and limits virus spread in transgenic plants. Plant J. 4: 959–970.
Lauber E., Bleykasten-Grosshans C., Erhardt M., Bouzoubaa S., Jonard G., Richards K.E. and Guilley H. 1998. Cell-to-cell movement of beet necrotic yellow vein virus. I. Heterologous complementation experiments provide evidence for specific interactions among the triple gene block proteins. Mol. Plant-Microbe Interact. 11: 618–625.
Lough T.J., Shash K., Xoconostle-Cazares B., Hofstra K.R., Beck D.L., Balmori E., Forster R.L.S. and Lucas W.J. 1998. Molecular dissection of the mechanism by which potexvirus triple gene block proteins mediate cell-to-cell transport of infectious RNA. Mol. Plant-Microbe Interact. 11: 801–814.
McGeachy K.D. and Barker H. 2000. Potato mop-top virus RNA can move long distance in the absence of coat protein: evidence from resistant, transgenic plants. Mol. Plant-Microbe Interact. 13: 125–128.
Powell-Abel P., Nelson R.S., De B., Hoffman N., Rogers S.G., Fraley R.T. and Beachy R.N. 1986. Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 232: 738–743.
Pringle C.R. 1999. Virus taxonomy - 1999. Arch. Virol. 144: 421–429.
Reavy B., Arif M., Kashiwazaki S., Webster K.D. and Barker H. 1995. Immunity to potato mop-top virus in Nicotiana benthamiana plants expressing the coat protein gene is effective against fungal inoculation of the virus. Mol. Plant-Microbe Interact. 8: 286–291.
Reavy B., Sandgren M., Barker H., Heino P. and Oxelfelt P. 1997. A coat protein transgene from a Scottish isolate of potato mop-top virus mediates strong resistance against Scandinavian isolates which have similar coat protein genes. Eur. J. Plant Path. 103: 829–834.
Rowhani A., Biardi L., Routh G. and Daubert S.D. 1998. Development of a sensitive colorimetric PCR assay for detection of virus in woody plants. Plant Dis. 8: 880–884.
Sambrook J., Fritsch E.F. and Maniatis T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Plainview, NY.
Sandgren M. 1995. Potato mop-top virus (PMTV): distribution in Sweden, development of symptoms during storage and cultivar trials in field and glasshouse. Potato Res. 38: 387–397.
Sandgren M. 1996. On spraing in potato. Ph.D. thesis, Swedish University of Agricultural Sciences, Uppsala.
Savenkov E.I., Sandgren M. and Valkonen J.P.T. 1999. Complete sequence of RNA 1 and the presence of tRNA-like structures in all RNAs of potato mop-top virus, genus Pomovirus. J. Gen. Virol. 80: 2779–2784.
Scott K.P., Kashiwazaki S., Reavy B. and Harrison B.D. 1994. The nucleotide sequence of potato mop-top virus RNA 2: a novel type of genome organization for a furovirus. J. Gen. Virol. 75: 3561–3568.
Seppänen P., Puska R., Honkanen J., Tyulkina L.G., Fedorkin O., Morozov S.Yu. and Atabekov J.G. 1997. Movement protein derived resistance to triple gene block-containing plant viruses. J. Gen. Virol. 78: 1241–1246.
Silva G.H., Chase R., Hammerschmidt M.L., Vitosh M.L. and Kitchen R.B. 1991. Irrigation, nitrogen and gypsum effects on specific gravity and internal defects on Atlantic potatoes. Am. Potato J. 68: 751–765.
Solovyev A.G., Savenkov E.I., Grdzelishvili V.Z., Kalinina N.O., Morozov S.Yu., Schiemann J. and Atabekov J.G. 1999. Movement of hordeivirus hybrids with exchanges in the triple gene block. Virology 253: 278–287.
Solovyev A.G., Stroganova T.A., Zamyatnin A.A., Fedorkin O.N., Schiemann J. and Morozov S.Yu. 2000. Subcellular sort-ing of small membrane-associated triple gene block proteins: TGBp3-assisted targeting of TGBp2. Virology 269: 113–127.
Visser R.G.F. 1991. Regeneration and transformation of potato by Agrobacterium tumefaciens. In: Lindsey K. (ed.), Plant Tissue Culture Manual, Kluwer Academic Publishers, Dordrecht, Netherlands. pp. B5/1–B5/9.
Visser R.G.F., Stolte A. and Jacobsen E. 1991. Expression of a chimaeric granule-bound starch synthase-GUS gene in transgenic potato plants. Plant Mol. Biol. 17: 691–699.
Walkerpeach C.R. and Velten J. 1994. Agrobacteriummediated gene transfer to plant cells: cointegrate and binary vector systems. In: Gelvin S.B. and Schilperoort R.A. (eds.), Plant Molecular Biology Manual, 2nd ed. Kluwer Academic Publishers, Dordrecht, Netherlands, pp. B1/1–B1/19.
Yang Y., Ding B., Baulcombe D.C. and Verchot J. 2000. Cell-to-cell movement of the 25K protein of potato virus X is regulated by three other viral proteins. Mol. Plant-Microbe Interact. 13: 599–605.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Melander, M., Lee, M. & Sandgren, M. Reduction of potato mop-top virus accumulation and incidence in tubers of potato transformed with a modified triple gene block gene of PMTV. Molecular Breeding 8, 197–206 (2001). https://doi.org/10.1023/A:1013719726848
Issue Date:
DOI: https://doi.org/10.1023/A:1013719726848