Abstract
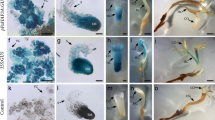
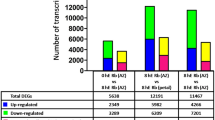
The aquatic fern Marsilea quadrifolia produces different types of leaves in response to changes in natural environment and culture conditions. When the conditions are in favor of producing the submerged-type leaves, exogenous application of the plant hormone abscisic acid (ABA) induces the formation of aerial-type leaves. Tissues responsive to ABA were localized to the shoot apical meristem and the associated organ primordia. From these tissues, at least two tiers of ABA-regulated early genes were identified, including seven primary genes and seventeen secondary genes. These genes, designated ABRH for ABA-responsive heterophylly, showed diverse expression patterns during the course of heterophyllous induction. Changes in the transcript level of ABRH genes started early, within 0.5–1.0 h after the addition of ABA to the culture medium. Some changes were transient while the others were persistent. The ABRHs contain extensive sequence homology to known genes, including those encoding transcription factors, protein kinases, membrane transporters, metabolic enzymes, structural proteins and those encoded by the chloroplast genome. Identification of these ABRHs is a first step toward the understanding of the regulation mechanisms of heterophylly, and the results suggest the involvement of novel metabolic and regulatory pathways in ABA-controlled morphogenesis.
Similar content being viewed by others
References
Aarts, M.G., Keijzer, C.J., Stiekema, W.J. and Pereira, A. 1995. Molecular characterization of the CER1 gene of Arabidopsis involved in epicuticular wax biosynthesis and pollen fertility. Plant Cell 7: 2115–2127.
Allsopp, A. 1952. Experimental and analytical studies of pteridophytes. XVII. the effect of various physiologically active substances on the development of Marsilea in sterile culture. Ann. Bot. 16: 165–183.
Allsopp, A. 1962. The effects of gibberellic acid on morphogenesis in Marsilea drummondii A. Br. Phytomorphol. 12: 1–10.
Allsopp, A. 1965. Land and water forms: physiological aspects. Handbuch Pflanzenphysiol. 15: 1236–1255.
Allsopp, A. 1967. Heteroblastic development in vascular plants. In: M. Abercrombie and J. Brachet (Eds.) Advances in Morphogenesis, Academic Press, New York, pp. 127–171.
Altschul, S.F., Madden, T.L., Schäffer, A.A., Zhang, J., Zhang, Z., Miller, W. and Lipman, D.J. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl. Acids Res. 25: 3389–3402.
Anderberg, R.J. and Walker-Simmon, M.K. 1992. Isolation of a wheat cDNA clone for an abscisic acid-inducible transcript with homology to protein kinases. Proc. Natl. Acad. Sci. USA 89: 10183–10187.
Baudo, M.M., Meza-Zepeda, L.A., Palva, E.T. and Heino, P. 1996. Induction of homologous low temperature and ABA-responsive genes in frost resistant (Solanum commersonii) and frost-sensitive (Solanum tuberosum cv. Bintje) potato species. Plant Mol. Biol. 30: 331–336.
Becraft, P.W., Stinard, P.S., McCarty, D.R. 1996. CRINKLY4: a TNFR-like receptor kinase involved in maize epidermal differentiation. Science 273: 1406–1409.
Busk, P.K and Pagès, M. 1998. Regulation of abscisic acid-induced transcripition. Plant Mol. Biol. 37: 425–435.
Chen, J., Dellaporta, S. 1994. Urea-based plant DNA miniprep. In: M. Freeling and V. Walbot (Eds.) The Maize Handbook, Springer-Verlag, New York, pp. 526–527.
Choi, H.I., Hong, J.H., Ha, J.O., Kang, J.Y. and Kim, S.Y. 2000. ABFs, a family of ABA-responsive element binding factors. J. Biol. Chem. 275: 1723–1730.
Deng, M.D., Peng, S. and Lemieux, B. 1996. Genomic (Accession No. X95964) and cDNA (Accession No. X95965) sequences of the CER1-like gene of Arabidopsis thaliana derived from a plant DNA/T-DNA insertion junction. (PGR96-019). Plant Physiol. 110: 1436.
Foster, R., Izawa, T. and Chua, N.-H. 1994. Plant bZIP proteins gather at ACGT elements. FASEB J. 8: 129–200.
Gaudet, J.J. 1963. Marsilea vestita: conversion of the water form to the land form by darkness and by far-red light. Science 140: 975–976.
Gaudet, J.J. 1964. Morphology of Marsilea vestita. II. Morphology of the adult land and submerged leaves. Am. J. Bot. 51: 495–502.
Gaudet, J.J. 1965. The effect of various environmental factors on the leaf form of the aquatic fern Marsilea vestita. Physiol. Plant. 18: 674–686.
Gifford, E.M. and Foster, A.S.1988. Morphology and Evolution of Vascular Plants. W. H. Freeman and Company, New York.
Hansen, J.D., Pyee, J., Xia, Y., Wen, T.J., Robertson, D.S., Ko-lattukudy, P.E., Nikolau, B.J. and Schnable, P.S. 1997. The glossy1 locus of maize and an epidermis-specific cDNA from Kleinia odora define a class of receptor-like proteins required for the normal accumulation of cuticular waxes. Plant Physiol. 113: 1091–1100.
Jepson, I., Bray, J., Jenkins, G., Schuch, W. and Edwards, K. 1991. A rapid procedure for the construction of PCR cDNA libraries from small amounts of plant tissue. Plant Mol. Biol. Rep. 9: 131–138.
Kerstetter, R.A. and Poethig, R.S. 1998. The specification of leaf identity during shoot development. Annu. Rev. Cell Dev. Biol. 14: 373–398.
Laetsch, W.M. and Briggs, W.R. 1961. Kinetin modification of sporeling ontogeny in Marsilea vestita. Am. J. Bot. 48: 369–377.
Liang, P. and Pardee, A.B. 1992. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 257: 967–971.
Lin, B.L and Tang, W.J. 1999. Blue light and abscisic acid independently induce heterophyllous switch in Marsilea quadrifolia. Plant Physiol. 119: 429–434.
Liu, B.L.L. 1984. Abscisic acid induces land form characteristics in Marsilea quadrifolia L. Am. J. Bot. 71: 638–644.
Lu, G., Paul, A.-L., McCarty, D.R. and Ferl, R.J. 1996. Transcription factor veracity: is GBF3 responsible for ABA-regulated expression of Arabidopsis Adh? Plant Cell 8: 847–857.
McCurdy, D.W. and Kim, M. 1998. Molecular cloning of a novel fimbrin-like cDNA from Arabidopsis thaliana. Plant Mol. Biol. 36: 23–31.
Nakagawa, H., Ohmiya, K. and Hattori, T. 1996. A rice bZIP protein, designated OSBZ8, is rapidly induced by abscisic acid. Plant J. 9: 217–227.
Purugganan, M.D., Rounsley, S.D., Schmidt, R.J. and Yanofsky, M.F. 1995. Molecular evolution of flower development: diversification of the plant MADS box regulatory gene family. Genetics 140: 345–356.
Riechmann, J.L. and Meyerowitz, E.M. 1997. MADS domain proteins in plant development. Biol. Chem. 378: 1079–1101.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Plainview, NY.
Sanger, F., Nicklen, S. and Coulson, A.R. 1977. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Sculthorpe, C.D. 1967. The Biology of Aquatic Vascular Plants. Arnold, London.
Smith, L.G. and Hake, S. 1992. The initiation and determination of leaves. Plant Cell 4: 1017–1027.
Steeves, T.A. and Sussex, I.M. 1989. Patterns in Plant Development. Cambridge University Press, Cambridge, UK.
Stoebe, B., Martin, W. and Kowallik, K.V. 1998. Distribution and nomenclature of protein-coding genes in 12 sequenced chloroplast genomes. Plant Mol. Biol. Rep. 16: 243–255.
Takahashi, T., Mu, J.H., Gasch, A. and Chua, N.H. 1998. Identification by PCR of receptor-like protein kinases from Arabidopsis flowers. Plant Mol. Biol. 37: 587–596.
Trachtulcova, P., Janatova, I., Kohlwein, S.D. and Hasek, J. 2000. Saccharomyces cerevisiae gene ISW2 encodes a microtubule-interacting protein required for premeiotic DNA replication. Yeast 16: 35–47.
Trewavas, A.J. and Jones, H.G. 1991. An assessment of the role of ABA in plant development. In: W.J. Davies. and H.G. Jones (Eds.) Abscisic Acid: Physiology and Biochemistry, BIOS Sci-entific Publishers, Oxford, pp. 169–188.
Wakasugi, T., Sugita, M., Tsudzuki, T. and Sugiura, M. 1998. Updated gene map of tobacco chloroplast DNA. Plant Mol. Biol. Rep. 16: 231–241.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hsu, TC., Liu, HC., Wang, JS. et al. Early genes responsive to abscisic acid during heterophyllous induction in Marsilea quadrifolia . Plant Mol Biol 47, 703–715 (2001). https://doi.org/10.1023/A:1013612331583
Issue Date:
DOI: https://doi.org/10.1023/A:1013612331583