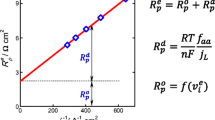
Abstract
We developed a method for determining the corrosion rate and Tafel constants on the basis of a two-electrode scheme for measuring polarization data in a nonlinear portion of the polarization curve with diffusion control of the cathode reaction. The corrosion current was calculated from two values of polarization currents i 1 and i 2 at the corresponding values of polarization ΔU 1 and ΔU 2 = 2ΔU 1 according to the formula
Using steel 20 in 0.1 M NaCl solution as an example, we calculated corrosion parameters, depending on polarization and the scanning rate of potential; the calculated values agree well with the results of massmetric and analytic evaluations of the corrosion rate. The comparison of the data obtained in the present work with those obtained by using the Engell and Beleevskii–Kudelin methods confirms advantages of the two-electrode measuring method, including its higher precision and simple instrumentation.
Similar content being viewed by others
References
S. Barnartt and M. Donaldson, “Threepoint method for evaluating corrosion current and Tafel slopes from a single polarization curve,” Corrosion, 39, No. 1, 33–35 (1983).
G. A. Rocchini, “A computerized tool for corrosion rate monitoring,” Corrosion, 43, No. 6, 326–331 (1987).
N. D. Green and R. H. Gandi, “Betacrunch version 2.0,” Mater. Perform., 26, No. 7, 52–53 (1987).
M. D. Reingeverts, N. A. Sergeeva, and N. G. Semenova, “Determination of kinetic parameters of metal corrosion from polarization data,” Zashch. Metal., 29, No. 2, 235–241 (1993).
F. Mansfeld, “Determination of corrosion current by the polarizationresistance method,” in: M. G. Fontana and R. W. Staehle (editors), Corrosion Science and Technology [Russian translation], Vol. 6, Metallurgiya, Moscow (1980), pp. 259–265.
M. J. Danielson, “An evaluation of the threepoint method to measure corrosion rate,” Corrosion, 38, No. 11, 580–586 (1982).
J. Jankowskii and R. Juchniewicz, “A fourpoint method for corrosion rate determination,” Cor. Sci., 20, No. 7, 841–851 (1980).
V. S. Beleevskii and Yu. I. Kudelin, “Calculation of corrosion rate and Tafel constants from two or three values of polarization current of the same sign near corrosion potential”, Zashch. Metal., 25, No. 1, 80–85 (1989).
S. V. Dudenkov, A. B. Shein, G. V. Khaldeev, and A. N. Syur, “Evaluation of the region of applicability of the Stern-Geary equation in computeraided calculation of corrosion rate using measurements of polarization,” Zashch. Metal., 19, No. 3, 500–502 (1983).
Yu. S. Gerasimenko, “Nonlinearity of a polarization curve and accuracy of polarizationresistance measurements,” Zashch. Metal., 15, No. 6, 673–677 (1979).
R. Bandy, “Measurement of corrosion rate using two electrodes,” Electrochim. Acta, 26, No. 1, 149–159 (1981).
Yu. S. Gerasimenko and N. A. Belousova, A Method for Determining the Corrosion Rate of Metals under Conditions of Diffusion Control and Passivation [in Russian], Author's Certificate No. 94076425 MKI: G 01 27/26, Promyslova Vlasnist'. Ofitsiinyi Byuleten'., No. 1, 292 (1996).
G. Rocchini, “The use of the threepoint method for evaluating corrosion rates,” Cor. Sci., 40, No. 6, 861–870 (1998).
G. Rocchini, “The analysis of the experimental data by Mansfeld's method,” Cor. Sci., 38, No. 7, 1019–1031 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Herasymenko, Y.S., Bilousova, N.A. Double Polarization Method for the Determination of Corrosion Rate under Conditions of Diffusion Control. Materials Science 37, 637–643 (2001). https://doi.org/10.1023/A:1013280922912
Issue Date:
DOI: https://doi.org/10.1023/A:1013280922912