Abstract
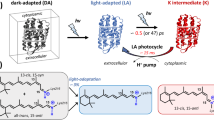
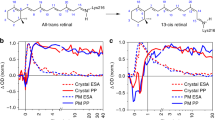
The primary events in the photosynthetic retinal protein bacteriorhodopsin (bR) are reviewed in light of photophysical and photochemical experiments with artificial bR in which the native retinal polyene is replaced by a variety of chromophores. Focus is on retinals in which the “critical” C13=C14 bond is locked with respect to isomerization by a rigid ring structure. Other systems include retinal oxime and non-isomerizable dyes noncovalently residing in the binding site. The early photophysical events are analyzed in view of recent pump–probe experiments with sub-picosecond time resolution comparing the behavior of bR pigments with those of model protonated Schiff bases in solution. An additional approach is based on the light-induced cleavage of the protonated Schiff base bond that links retinal to the protein by reacting with hydroxylamine. Also described are EPR experiments monitoring reduction and oxidation reactions of a spin label covalently attached to various protein sites. It is concluded that in bR the initial relaxation out of the Franck–Condon (FC) state does not involve sub-stantial C13=C14 torsional motion and is considerably catalyzed by the protein matrix. Prior to the decay of the relaxed fluorescent state (FS or I state), the protein is activated via a mechanism that does not require double bond isomerization. Most plausibly, it is a result of charge delocalization in the excited state of the polyene (or other) chromophores. More generally, it is concluded that proteins and other macromolecules may undergo structural changes (that may affect their chemical reactivity) following optical excitation of an appropriately (covalently or non-covalently) bound chromophore. Possible relations between the light-induced changes due to charge delocalization, and those associated with C13=C14 isomerization (that are at the basis of the bR photocycle), are discussed. It is suggested that the two effects may couple at a certain stage of the photocycle, and it is the combination of the two that drives the cross-membrane proton pump mechanism.
Similar content being viewed by others
REFERENCES
Ottolenghi, M., and Sheves, M. (eds.) (1995) The Photophysics and Photochemistry of Retinal Proteins. Isr. J. Chem., 35, 193–515.
Lanyi, J. (ed.) (2000) Biochim. Biophys. Acta Bioenerg., 1460, 1–239.
Haupt, U., Tittor, J., and Oesterhelt, D. (1999) Ann. Rev. Biophys. Biomol. Struct., 21, 367–399.
Kochendoerfer, G. G., and Mathies, R. A. (1995) Isr. J. Chem., 35, 211–226.
Stuart, J. A., and Birge, R. R. (1996) Biomembranes, 2A, 33–139.
Rosenfeld, T., Honig, B., Ottolenghi, M., Hurley, J., and Ebrey, T. G. (1977) Pure Appl. Chem., 49, 341–351.
Hurley, J., Ebrey, T. G., Honig, B., and Ottolenghi, M. (1977) Nature (London), 270, 540–542.
Sharkov, S., Pakulev, A., Chekalin, S., and Matveetz, Y. (1985) Biochim. Biophys. Acta, 808, 94–102.
Mathies, R. A., Brito-Cruz, C. H., Pollard, T. W., and Shank, C. V. (1988) Science, 240, 777–779.
Dobler, J., Zinth, W., Kaiser, K., and Oesterhelt, D. (1988) Chem. Phys., 144, 215–220.
Kobayashi, T., Terauchi, M., Kouyama, T., Yoshizawa, M., and Taiji, M. (1990) SPIE, 1403, 407–416.
Kobayashi, T., Kim, M., Taiji, M., Iwasa, T., Nakagawa, M., and Tsuda, M. (1998) J. Phys. Chem. B., 102, 272–280.
Gai, F., Hasson, H. C., McDonald Kooper, J., and Anfinrud, P. A. (1998) Science, 279, 1886–1891.
Arlt, T., Schmidt, S., Zinth, W., Haupts, U., and Oesterhelt, D. (1995) Chem. Phys. Lett., 241, 559–565.
Kandori, H., Yoshihara, K., Tomioka, H., and Sasabe, H. (1992) J. Phys. Chem., 96, 6066–6071.
Loppnow, G. R., and Mathies, R. A. (1988) Biophys. J., 54, 35–43.
Palings, I., Pardoen, J. A., van den Berg, E., Winkel, C., Lugetenburg, J., and Mathies, R. A. (1987) Biochemistry, 26, 2544–2556.
Hasson, K. C., Gai, F., and Anfinrud, P. A. (1996) Proc. Natl. Acad. Sci. USA, 93, 16124–16129.
Humphrey, W., Lu, H., Logunov, I., Werner, H., and Schulten, K. (1998) Biophys. J., 75, 1689–1699.
Gonzalez-Luque, R., Garavelli, M., Bernardi, F., Merchan, M., Robb, M., and Olivucci, M. (2000) Proc. Natl. Acad. Sci. USA, 17, 9379–9384.
Salem, L., and Bruckmann, P. (1975) Nature (London), 258, 526–529.
Lewis, A. (1978) Proc. Natl. Acad. Sci. USA, 75, 543–547.
Xu, D., Martin, D., and Schulten, K. (1996) Biophys. J., 70, 453–460.
Ottolenghi, M., and Sheves, M. (1989) J. Membr. Biol., 112, 193–212.
Nakanishi, K., and Crouch, R. (1995) J. Isr. Chem., 35, 253–272.
Haran, G., Wynne, K., Xie, A., He, Q., Chance, M., and Hochstasser, R. M. (1996) Chem. Phys. Lett., 26, 389–395.
Gai, G., McDonald, P. A., and Anfinrud, P. A. (1997) J. Am. Chem. Soc., 119, 6201–6202.
Zhong, Q., Ruhman, S., Ottolenghi, M., Sheves, M., Friedman, N., Atkinson, G. H., and Delaney, J. K. (1996) J. Am. Chem. Soc., 118, 12828–12829.
Ye, T., Friedman, N., Gat, Y., Atkinson, G., Sheves, M., Ottolenghi, M., and Ruhman, S. (1999) J. Phys. Chem. B, 103, 5122–5130.
Akiyama, R., Yoshimori, A., Kakitani, T., Imamoto, Y., Shichida, Y., and Hatamo, Y. (1997) J. Phys. Chem. A, 101, 412–417.
Myers, A., Harris, R., and Mathies, R. (1983) J. Phys. Chem., 79, 603–613.
Garavelli, M., Celani, P., Bernardi, F., Robb, M. A., and Olivucci, M. (1997) J. Am. Chem. Soc., 119, 6891–6901.
Song, L., and El-Sayed, M. (1998) J. Am. Chem. Soc., 120, 8889–8890.
Govindjee, R., Balashov, V. S., and Ebrey, T. (1990) Biophys. J., 58, 597–608.
Balashov, S. P., Karneeva, N. V., Litvin, F. F., and Sineshchekov, V. A. (1987) in Retinal Proteins (Ovchinnikov, Yu. A., ed.) VNU Science Press, Utrecht, The Netherlands, pp. 505–517.
Dexheimer, S. L., Wang, Q., Peteanu, L. A., Pollard, W. T., Mathies, R. A., and Shank, C. V. (1992) Chem. Phys. Lett., 188, 61–67.
Wang, Q., Schoenlein, R., Peteanu, L., Mathies R., and Shank, C. (1994) Science, 266, 422–424.
Althaus, T., Weisfeld, R., Lohrmann, M., and Stockburger, M. (1995) Isr. J. Chem., 35, 227–251.
Ye, T., Gershgoren, E., Friedman, N., Ottolenghi, M., Sheves, M., and Ruhman, S. (1999) Chem Phys., 314, 429–434.
Garavelli, M., Negri, F., and Olivucci, M. (1999) J. Am. Chem. Soc., 121, 1023–1029.
Kandori, H., and Sasabe, H. (1993) Chem. Phys. Lett., 216, 126–132.
Kandori, H., Katsuta, Y., Ito, M., and Sasabe, H. (1995) J. Am. Chem. Soc., 117, 2669–2670.
Logunov, S. L., Song, L., and El-Sayed, M. A. (1996) J. Phys. Chem., 100, 18586–18591.
Hamm, P., Zurek, M., Röschinger, T., Patzelt, H., Oesterhelt, D., and Zinth, W. (1996) Chem. Phys. Lett., 263, 613–621.
Hou, B., Friedman, N., Ruhman, S., Sheves, M., and Ottolenghi, M. (2001) J. Phys. Chem., in press.
Rousso, I., Khachatryan, E., Gat, Y., Brodsky, I., Ottolenghi, M., Sheves, M., and Lewis, A. (1997) Proc. Natl. Acad. Sci. USA, 94, 7937–7941.
Losi, A., Michler, J., Gartner, W., and Braslavsky, S. (2000) Photochem. Photobiol., 72, 590–597.
Oesterhelt, D., Meetzen, M., and Schumann, L. (1973) Eur. J. Biochem., 40, 453–463.
Oesterhelt, D., Shumann, L., and Gruber, H. (1974) FEBS Lett., 44, 257–261.
Subramaniam, S., Marti, T., Rösselet, S. J., Rothschild, K. J., and Khorana, H. G. (1991) Proc. Natl. Acad. Sci. USA, 88, 2583–2587.
Rousso, I., Gat, Y., Lewis, A., Sheves, M., and Ottolenghi, M. (1998) Biophys. J., 75, 413–417.
Aharoni, A., Weiner, L., Ottolenghi, M., and Sheves, M. (2000) J. Biol. Chem., 275, 21010–21016.
Feix, Y. B., and King, C. (1998) Spin-Labeling, Plenum Press, New York, pp. 251–281.
Hubbell, W., Gross, A., Langen, R., and Leitzow, M. (1998) Curr. Opin. Struct. Biol., 8, 649–656.
Altenbach, C., Fitsch, L., Khorana, H. G., and Hubbell, W. (1989) Biochemistry, 28, 7806–7812.
Altenbach, C., Marti, T., Khorana, H. G., and Hubbell, W. (1990) Science, 248, 1088–1092.
Steinhoff, H., Mollaaghababa, R., Altenbach, C., Hideg, K., Krebs, M., Khorana, H. G., and Hubbell, W. (1994) Science, 266, 105–107.
Hubbell, W., and Altenbach, C. (1994) Curr. Opin. Struct. Biol., 4, 566–573.
Thorgeirsson, T., Xiao, W., Brown, L., Needleman, R., Lanyi, J., and Shin, Y. (1997) J. Mol. Biol., 273, 951–957.
Rink, T., Riesk, J., Oesterhelt, D., Gerwert, K., and Steinhoff, H. (1997) Biophys. J., 73, 983–993.
Pfeiffer, M., Rink, T., Gerwert, K., Oesterhelt, D., and Steinhoff, H. (1999) J. Mol. Biol., 275, 163–171.
Aharoni, A., Weiner, L., Ottolenghi M., and Sheves, M. (2001) J. Am. Chem. Soc., 123, 6612–6616.
Huang, J., Chen, Z., and Lewis, A. (1989) J. Phys. Chem., 93, 3314–3320.
Birge, R., and Zheng, X. (1990) J. Chem. Phys., 94, 7178–7179.
Clays, K., Hendrick, E., Triest, M., Verhiest, T., Persoons, A., Dehu, C., and Bredas, J. (1993) Science, 262, 1419–1422.
Birge, R., and Zhang, C. (1990) J. Chem. Phys., 92, 7178–7195.
Atkinson, G. H., Ujj, L., and Zhou, Y. (2000) J. Phys. Chem., 104, 4130–4139.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aharoni, A., Hou, B., Friedman, N. et al. Non-Isomerizable Artificial Pigments: Implications for the Primary Light-Induced Events in Bacteriorhodopsin. Biochemistry (Moscow) 66, 1210–1219 (2001). https://doi.org/10.1023/A:1013175000873
Issue Date:
DOI: https://doi.org/10.1023/A:1013175000873