Abstract
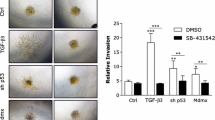
We have recently shown that either exogenous or endogenous, transfected OPN induces both uPA expression and increased invasiveness of 21PT (non-tumorigenic) and 21NT (tumorigenic) human mammary epithelial cells. Here we asked whether uPA contributes functionally to the increased invasiveness of these cells. The most invasive OPN-transfected cells were assessed for migration through Matrigel in transwell assays, in the presence or absence of various blocking antibodies and uPA inhibitors. Antibodies to both uPA and uPA receptor (uPAR) were shown to significantly inhibit cell invasion, as did the uPA inhibitors (plasminogen activator inhibitor-1 [PAI-1], p-aminobenzamidine [PABN], aprotinin, and amiloride). Both anti-uPA and anti-uPAR antibodies inhibited invasion to a level comparable to that of the control vector transfected cells. In contrast, non-specific IgG showed no anti-invasive effect. Cell migration experiments performed with the parental cell lines in the presence or absence of anti-uPA or anti-uPAR antibodies showed that uPA is also required for migratory responsiveness to exogenous OPN. These data thus provide direct evidence that OPN-induced invasion and migration of these cells requires uPA.
Similar content being viewed by others
References
Tuck AB, O'Malley FP, Singal H, Harris JF, Tonkin KS, Kerkvliet N, Saad Z, Doig G, Chambers AF: Osteopontin expression in a group of lymph node negative breast cancer patients. Int J Cancer 79: 502-508, 1998
Singhal H, Bautista DS, Tonkin KS, O'Malley FP, Tuck AB, Chambers AF, Harris JF: Elevated plasma osteopontin inmetastatic breast cancer associated with increased tumor burden and decreased survival. Clin Cancer Res 3: 605-611, 1997
Tuck AB, Arsenault DM, O'Malley FP, Hota C, Ling MC, Wilson SM, Chambers AF: Osteopont in induces increased invasiveness and plasminogen activator expression of human mammary epithelial cells. Oncogene 18: 4237-4246, 1999
Baricos WH, Cortez SL, el-Dahr SS, Schnaper HW: ECM degradation by cultured human mesangial cells is mediated by a PA/plasmin/MMP-2 cascade. Kidney Int 47: 1039-1047, 1995
Mazzieri R, Masiero L, Zanetta L, Monea S, Onisto M, Garbisa S, Mignatti P: Control of type IV collagenase activity by components of the urokinase-plasmin system: a regulatory mechanism with cell-bound reactants. EMBO J 16: 2319-2332, 1997
Werb Z: ECM and cell surface proteolysis: regulating cellular ecology. Cell 91: 439-442, 1997
Andreasen P-A, Kjøller L, Christensen L, Duffy MJ: The urokinase-type plasminogen activator system in cancer metastasis: a review. Int J Cancer 72: 1-22, 1997
Yebra M, Parry GCN, Strömblad S, Mackman N, Rosenberg S, Mueller BM, Cheresh DA: Requirement of receptorbound urokinase-type plasminogen activator for integr in ?v? 5-directed cell migration. J Biol Chem 271: 29393-29399, 1996
Carriero MV, Vecchio SD, Capozzoli M, Franco P, Fontana L, Zannetti A, Botti G, D'Aiuto G, Salvatore M, Stoppelli MP: Urokinase receptor interacts with ?v?5 vitronectin receptor, promoting urokinase-dependent cell migration in breast cancer. Cancer Res 59: 5307-5314, 1999
Band V, Zajchowski D, Swisshelm K, Trask D, Kulesa V, Cohen C, Connolly J, Sager R: Tumor progression in four mammary epithelial cell lines derived from the same patient. Cancer Res 50: 7351-7357, 1990
Tuck AB, Wilson SM, Chambers AF: Ras transfection and expression does not induce progression from tumorigenicity to metastatic ability in mouse LTA cells. Clin Exp Metast 8: 417-431, 1990
Tuck AB, Wilson SM, Khokha R, Chambers AF: Different patterns of gene expression in ras-resistant and ras-sensitive cells. J Natl Cancer Inst 83: 485-491, 1991
Young MF, Kerr JM, Termine JD, Wewer UM, Wang MG, McBride OW, Fisher LW: cDNA cloning, mRNA distribution and heterogeneity, chromosomal location, and RFLP analysis of human osteopontin (OPN). Genomics 7: 491-502, 1990
Riccio A, Grimaldi G, Verde P, Sebastio G, Boast S, Blasi F: The human urokinase-plasminogen activator gene and its promoter. Nucl Acids Res 13: 2759-2771, 1985
Roldan AL, Cubellis MV, Masucci MT, Behrendt N, Lund LR, Dano K, Appella E, Blasi F: Cloning and expression of the receptor for human urokinase plasminogen activator, a central molecule in cell surface, plasmin dependent proteolysis. EMBO J 9: 467-474, 1990
Simon C Juarez J, Nicolson GL, Boyd D: Effect of PD 098059, a specific inhibitor of mitogen-activated protein kinase, on urokinase expression and in vitro invasion. Cancer Res 56: 5369-5374, 1996
Xuan JW, Hota C, Shigeyama Y, D'Errico JA, Somerman MJ, Chambers AF: Site-directed mutagenesis of the arginineglycine-aspartic acid sequence in osteopontin destroys cell adhesion and migration functions. J Cell Biochem 57: 680-690, 1995
Xuan JW, Hota C, Chambers AF: Recombinant GST-human osteopontin fusion protein is functional in RGD-dependent cell adhesion. J Cell Biochem 54: 247-255, 1994
Fisher JL, Field CL, Zhou H, Harris TL, Henderson MA, Choong PF: Urokinase plasminogen activator system gene expression is increased in human breast carcinoma and its bone metastases-a comparison of normal breast tissue, non-invasive and invasive carcinoma and osseous metastases. Breast Cancer Res Treat 61: 1-12, 2000
Senger DR, Perruzzi CA: Cell migration promoted by a potent GRGDS-containing thrombin-cleavage fragment of osteopontin. Biochim Biophys Acta 1314: 13-24, 1996
Tuck AB, Elliott BE, Hota C, Tremblay E, Chambers AF: Osteopontin-induced, integrin-dependent migration of human mammary epithelial cells involves activation of the hepatocyte growth factor receptor (Met). J Cell Biochem 78: 465-475, 2000
Zohar R, Suzuki N, Suzuki K, Arora P, Glogauer M, McCulloch CA, Sodek J: Intracellular osteopont in is an integral component of the CD44-ERM complex involved in cell migration. J Cell Physiol 184: 118-130, 2000
Sung V, Gilles C, Murray A, Clarke R, Aaron AD, Azumi N, Thompson EW: The LCC15-MB human breast cancer cell line expresses osteopontin and exhibits an invasive and metastatic phenotype. Exp Cell Res 241: 273-284, 1998
Oates AJ, Barraclough R, Rudland PS: The identification of osteopont in as a metastasis-related gene product in a rodent mammary tumour model. Oncogene 13: 97-104, 1996
Chen H, Ke Y, Oates AJ, Barraclough R, Rudland PS: Isolation of and effector for metastasis-inducing DNAs from a human metastatic carcinoma cell line. Oncogene 14: 1581-1588, 1997
Hu DD, Lin EC, Kovach NL, Hoyer JR, Smith JW: A biochemical characterization of the binding of osteopontin to integrins alpha v beta 1 and alpha v beta5. J Biol Chem 270: 26232-26238, 1995
Liaw L, Skinner MP, Raines EW, Ross R, Cheresh DA, Schwartz SM, Giachelli CM: The adhesive and migratory effects of osteopont in are mediated via distinct cell surface integrins. role of alpha v beta 3 in smooth muscle cell migration to osteopontin in vitro. J Clin Invest 95: 713-724, 1995
Smith LL, Cheung HK, Ling LE, Chen J, Sheppard D, Pytela R, Giachelli CM: Osteopont in N-terminal domain contains a cryptic adhesive sequence recognized by alpha9 beta1 integrin. J Biol Chem 271: 28485-28491, 1996
Denda S, Reichardt LF, Muller U: Identification of osteopontin as a novel ligand for the integrin alpha8 beta1 and potential roles for this integrin-ligand interaction in kidney morphogenesis. Mol Biol Cell 9: 1425-1435, 1998
Bayless KJ, Meininger GA, Scholtz JM, Davis GE: Osteopontin is a ligand for the alpha4 beta1 integrin. J Cell Sci 111: 1165-1174, 1998
Weber GF, Ashkar S, Glimcher MJ, Cantor H: Receptor-ligand interaction between CD44 and osteopontin (Eta-1). Science 271: 509-512, 1996
Bourguignon LY, Gunja-Smith Z, Iida N, Zhu HB, Young LJ, Muller WJ, Cardiff RD: CD44v(3,8-10) is involved in cytoskeleton-mediated tumor cell migration and matrix metalloproteinase (MMP-9) association in metastatic breast cancer cells. J Cell Physiol 176: 206-215, 1998
Katagiri YU, Sleeman J, Fujii H, Herrlich P, Hotta H, Tanaka K, Chikuma S, Yagita H, Okumura K, Murakami M, Saiki I, Chambers AF, Uede T: CD44 variants but not CD44s cooperate with beta1-containing integrins to permit cells to bind to osteopontin independently of arginine-glycine-aspartic acid, thereby stimulating cell motility and chemotaxis. Cancer Res 59: 219-226, 1999
Jeffers M, Rong S, Vande Woude GF: Enhanced tumorigenicity and invasion-metastasis by hepatocyte growth factor/scatter factor-met signalling in human cells concomitant with induction of the urokinase proteolysis network. Mol Cell Biol 16: 1115-1125, 1996
Pepper MS, Matsumoto K, Nakamura T, Orci L, Montesano R: Hepatocyte growth factor increases urokinase-type plasminogen activator (u-PA) and u-PA receptor expression in Madin-Darby canine kidney epithelial cells. J Biol Chem 267: 20493-20496, 1992
Ried S, Jäger C, Jeffers M, Vande Woude GF, Graeff H, Schmitt M, Lengyel E: Activation mechanisms of the urokinase-type plasminogen activator promoter by hepatocyte growth factor/scatter factor. J Biol Chem 274: 16377-16386, 1999
Naldini L, Tamagnone L, Vigna E, Sachs M, Hartmann G, Birchmeier W, Daikuhara Y, Tsubouchi H, Blasi F, Comoglio PM: Extracellular proteolytic cleavage by urokinase is required for activation of hepatocyte growth factor/scatter factor. EMBO J 11: 4825-4833, 1992
Webb CP, Hose CD, Koochekpour S, Jeffers M, Oskarsson M, Sausville E, Monks A, Vande Woude GF: The geldanamycins are potent inhibitors of the hepatocyte growth factor/scatter factor-met-urokinase plasminogen activator-plasmin proteolytic network. Cancer Res 60: 342-349, 2000
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tuck, A.B., Hota, C. & Chambers, A.F. Osteopontin(OPN)-induced increase in human mammary epithelial cell invasiveness is urokinase (uPA)-dependent. Breast Cancer Res Treat 70, 197–204 (2001). https://doi.org/10.1023/A:1013095329825
Issue Date:
DOI: https://doi.org/10.1023/A:1013095329825