Abstract
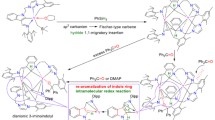
Germanium-containing organophosphorus betaine Et3P+—CHMe—GeMe2—S– was synthesized by the reaction of hexamethylcyclotrigermatrithiane with Et3P=CHMe. The structure of the betaine was established by X-ray diffraction analysis and multinuclear NMR spectroscopy. In the crystal, the P+—C—Ge—S– main chain of the molecule adopts a folded cis-gauche conformation due to strong Coulomb interactions between the anionic and cationic centers. The equilibrium geometry of the isolated molecule was calculated within the framework of the density functional theory (the PBE functional, the TZ2P basis set). The calculated geometric characteristics are in qualitative agreement with the X-ray data. The structure of the betaine is compared with the structure of its silicon-containing analog studied previously.
Similar content being viewed by others
References
Yu. A. Ustynyuk, M. S. Nechaev, D. N. Laikov, I. V. Borisova, N. N. Zemlyanskii, and V. N. Khrustalev, Izv. Akad. Nauk, Ser. Khim., 2001, 739 [Russ. Chem. Bull., Int. Ed., 2001, 50, 771 (Engl. Transl.)].
R. Tacke, Adv. Organomet. Chem., 1998, 46, 51.
G. Erker, Eur. J. Inorg. Chem., 2000, 1671.
I. V. Borisova, N. N. Zemlyansky, V. K. Belsky, N. D. Kolosova, A. N. Sobolev, Yu. N. Luzikov, Yu. A. Ustynyuk, and I. P. Beletskaya, J. Chem. Soc., Chem. Commun., 1982, 1090.
I. V. Borisova, N. N. Zemlyanskii, Yu. N. Luzikov, Yu. A. Ustynyuk, V. K. Belsky, N. D. Kolosova, M. M. Shtern, and I. P. Beletskaya, Dokl. Akad. Nauk SSSR, 1983, 269, 369 [Dokl. Chem., 1983, 269, 90 (Engl. Transl.)].
I. V. Borisova, N. N. Zemlyanskii, Yu. A. Ustynyuk, I. P. Beletskaya, and E. A. Chernyshev, Metalloorg. Khim., 1992, 5, 548 [Russ. Organomet. Chem., 1992, 5, 262 (Engl. Transl.)].
V. N. Khrustalev, V. N. Zemlyanskii, I. V. Borisova, Yu. A. Ustynyuk, and E. A. Chernyshev, Izv. Akad. Nauk, Ser. Khim., 2000, 922 [Russ. Chem. Bull., Int. Ed., 2000, 49, 920].
I. V. Borisova, N. N. Zemlyanskii, A. K. Shestakova, V. N. Khrustalev, Yu. A. Ustynyuk, and E. A. Chernyshev, Izv. Akad. Nauk, Ser. Khim., 2000, 935 [Russ. Chem. Bull., Int. Ed., 2000, 49, 933].
I. V. Borisova, N. N. Zemlyanskii, A. K. Shestakova, V. N. Khrustalev, Yu. A. Ustynyuk, and E. A. Chernyshev, Izv. Akad. Nauk, Ser. Khim., 2000, 1594 [Russ. Chem. Bull., Int. Ed., 2000, 49, 1583].
F. Bonhomme and M. G. Kanatzidis, Chemistry of Materials, 1998, 10, 1153.
O. M. Yaghi, Z. Sun, D. A. Richardson, and T. L. Groy, J. Am. Chem. Soc., 1994, 116, 807.
D. M. Nellis, Y. Ko, K. Tan, S. Koch, and J. B. Parise, Chem. Commun., 1995, 541.
M. S. Nechaev, I. V. Borisova, N. N. Zemlyanskii, D. N. Laikov, and Yu. A. Ustynyuk, Izv. Akad. Nauk, Ser. Khim., 2000, 1850 [Russ. Chem. Bull., Int. Ed., 2000, 49, 1823].
SMART and SAINT, Release 5.0. Area Detector Control and Integration Software, Bruker AXS, Analytical X-Ray Instruments, Madison, Wisconsin, USA, 1998.
G. M. Sheldrick, SHELXTL, V5.10, Bruker AXS Inc., Madison, WI-53719, USA, 1997.
D. N. Laikov, Chem. Phys. Lett., 1997, 281, 151.
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865.
K. A. Kocheshkov, N. N. Zemlyanskii, N. I. Sheverdina, and E. N. Panov, Metody elementoorganicheskoi khimii. Germanii. Olovo. Svinets [Methods of Organoelement Chemistry. Germanium. Tin. Lead], Nauka, Moscow, 1968, 105 pp. (in Russian).
E. G. Rochov, J. Am. Chem. Soc., 1948, 70, 1801.
R. Köster, D. Simie, and M. A. Grassberger, Justus Liebigs Ann. Chem., 1970, 739, 211.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Borisova, I.V., Zemlyanskii, N.N., Khrustalev, V.N. et al. Heteroorganic betaines. 7. Synthesis and structure of a germanium-containing organophosphorus betaine Et3P+—CHMe—GeMe2—S–. Russian Chemical Bulletin 50, 1679–1682 (2001). https://doi.org/10.1023/A:1013011324246
Issue Date:
DOI: https://doi.org/10.1023/A:1013011324246