Abstract
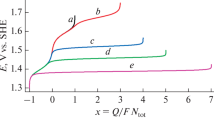
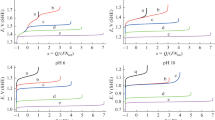
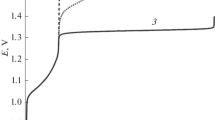
Products theoretically obtainable by electrochemical treatment of sodium chloride solutions in the dynamic mode are considered. Hydrated chlorine, and hypochlorous and hydrochloric acids are determined quantitatively by iodometric titration, spectrophotometry, and potentiometry. A comparative analysis is made of theoretical and experimental results obtained in determining chlorine-containing ions.
Similar content being viewed by others
REFERENCES
Spravochnik po elektrokhimii (Handbook of Electrochemistry), Sukhotin, A.M., Ed., Leningrad: Khimiya, 1991.
Dobos, D., Electrochemical Data, Budapest: Akad. Kiado, 1978.
Eigen, M. and Kustin, K., J. Am. Chem. Soc., 1962, vol. 84, no. 8, pp. 1355–1361.
Yakimenko, L.M., Proizvodstvo khlora, kausticheskoi sody i neorganicheskikh khlorproduktov (Manufacture of Chlorine, Caustic Soda, and Inorganic Chlorine Products), Moscow: Khimiya, 1974.
Cotton, F.A. and Wilkinson, G., Advanced Inorganic Chemistry, New York: Interscience, 1966.
Connick, R.E. and Chia, X.T., J. Am. Chem. Soc., 1959, vol. 81, no. 6, pp. 1280–1284.
Williams, W.J., Handbook of Anion Determination, London: Butterworths, 1979.
Karyakin, Yu.V. and Angelov, I.N., Chistye khimicheskie veshchestva (Pure Chemical Substances), Moscow: Khimiya, 1974.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Romanova, R.G., Agadzhanyan, S.I., Korshin, G.V. et al. Dynamics of Changes in the Anionic Composition of Sodium Chloride Solutions in a Flow-through Electrolyzer. Russian Journal of Applied Chemistry 74, 742–745 (2001). https://doi.org/10.1023/A:1012728630834
Issue Date:
DOI: https://doi.org/10.1023/A:1012728630834