Abstract
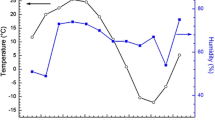
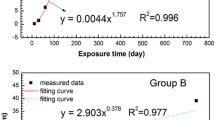
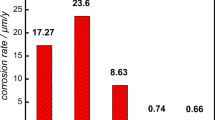
The corrosion of a carbon steel was studied in different atmospheres at sites in the Republic of Panama. The weight loss (corrosion penetration) suffered by the carbon steel is related to time by a bilogarithmic law. Mössbauer spectroscopy indicated the rust was composed of non-stoichiometric magnetite (Fe3-xO4), maghemite (γ-Fe2O3), goethite (α-FeOOH) of intermediate particle size, lepidocrocite (γ-FeOOH) and superparamagnetic particles. Magnetite formation is related to the alternating dry--wet cycles. Goethite is related to corrosion penetration by a saturation type of behavior, following a Langmuir type of relationship. Goethite in rust protects steel against further atmospheric corrosion.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jaén, J.A., de Villalaz, M.S., de Araque, L. et al. Kinetics and structural studies of the atmospheric corrosion of carbon steels in Panama. Hyperfine Interactions 110, 93–99 (1997). https://doi.org/10.1023/A:1012606928682
Issue Date:
DOI: https://doi.org/10.1023/A:1012606928682