Abstract
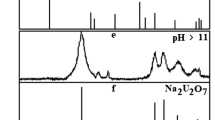
Interaction of U(VI) with Al(III) in solutions at pH ≥ 2 and in precipitates obtained at pH ≥ 5 was studied using spectrophotometry, luminescence, and IR spectroscopy. It was shown that in the range of pH 3–4, hydrolyzed forms of uranyl and of aluminium come into interaction. The mixed hydroxoaqua complexes (H2O)3UO2(μ-OH)2Al(H2O)3+ 4,(H2O)3UO2(μ-OH)2Al(OH)(H2O)2+ 3, or (H2O)4UO2OAl(OH)(H2O)2+ 4are likely to form in the solution. With an increase in pH, mixed polymers of a large size (oligomers) can form which further take part in the precipitate formation. The reaction between U(VI) and Al(III) in precipitates is confirmed by the data of IR spectroscopy and by the changes in the physico-chemical properties of these precipitates as compared with the properties of a mechanical mixture of separately precipitated uranium and aluminium. The important role of the oligomeric mixed forms in the formation of precipitate remains at pH levels varying from 5 to 14.
Similar content being viewed by others
REFERENCES
Yusov, A.B., Budantseva, N.A., Anan'ev, A.V., and Fedoseev, A.M., Radiokhimiya, 2000, vol. 42, no. 5, p. 413.
Shatalov, V.V., Tisov, Yu.I., Molchanov, S.V., and Smolina, I.V., Khimiya urana (Chemistry of Uranium), Laskorin, B.I., Ed., Moscow: Nauka, 1989, p. 434.
Kato, Y., Meinrath, G., Kimura, T., and Yoshida, Z., Radiochim. Acta, 1994, vol. 64, no. 2, p. 107.
Meinrath, G. and Schweinberger, M., Radiochim. Acta, 1996, vol. 75, no. 4, p. 205.
Meinrath, G., Radiochim. Acta, 1997, vol. 77, no. 4, p. 221.
Meinrath, G., Kato, Y., Kimura, T., and Yoshida, Z., Radiochim. Acta, 1998, vol. 82, p. 115.
Dai, S., Burleigh, M.C., Simonson, J.M., et al., Radiochim. Acta, 1998, vol. 81, no. 4, p. 195.
Petrosyants, S.P. and Buslaev, Yu.A., Zh. Neorg. Khim., 1999, vol. 44, no. 11, p. 1766.
Martell, A.E., Motekaitis, R.J., and Smith, R.M., Polyhedron, 1990, vol. 9, no. 2/3, p. 171.
Taylor, M.G. and Brothers, P.G., Chemistry of Aluminium, Gallium, Indium, and Thallium, Downs, A.J., Ed., London: Blackie Academic and Professional, 1993, p. 111.
Turner, R.C., Can. J. Chem., 1975, vol. 53, no. 19, p. 2811.
Turner, R.C., Can. J. Chem., 1976, vol. 54, no. 10, p. 1528.
Rowsell, J. and Nazar, L.F., J. Am. Chem. Soc., 2000, vol. 122, no. 15, p. 3777.
Satoh, I. and Choppin, G., Radiochim. Acta, 1992, vol. 56, no. 2, p. 85.
Jensen, M.P. and Choppin, G.R., Radiochim. Acta, 1998, vol. 82, p. 83.
Hrnecek, E. and Irlweck, K., Radiochim. Acta, 1999, vol. 87, nos. 1-2, p. 29.
Moll, H., Geipel, G., Brendler, V., et al., J. Alloys Compd., 1998, vols. 271–273, p. 765.
Meinrath, G., Lis, S., Stryla, Z., and Noubactep, C., J. Alloys Compd., 2000, vols. 300–301, p. 107.
Moolenaar, R.J., Evans, J.C., and McKeever, L.D., J. Phys. Chem., 1970, vol. 74, no. 20, p. 3629.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yusov, A.B., Budantseva, N.A., Fedoseev, A.M. et al. Interaction of Aluminium(III) with Uranyl Ions in the Course of Joint Hydrolysis. Russian Journal of Coordination Chemistry 27, 776–782 (2001). https://doi.org/10.1023/A:1012510904796
Issue Date:
DOI: https://doi.org/10.1023/A:1012510904796