Abstract
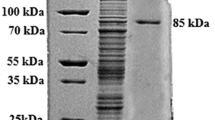
Two novel endo-type polygalacturonases (PGase), with molecular weights of 36 kDa and 40 kDa (named p36 and p40, respectively), were purified from the supernatant of the culture medium of a deep-sea yeast, strain N6, isolated from the Japan Trench. The N-terminal 20 amino acids of p36 and p40 were identical, and the sequence homology was 47.4% in comparison with the PGase of Fusarium moniliforme. A treatment of p40 with glycopeptidase F reduced the molecular weight to 36 kDa, suggesting that p40 possessed N-acetylglucosamines on its asparagine residues and p40 might be matured by glycosylation of p36.
Similar content being viewed by others
References
Barnby FM, Morpeth FF, Pyle DL (1990) Endopolygalacturonase production from Kluyveromyces marxianus. I. Resolution, purification and partial characterization of the enzyme. Enzyme Microb. Technol. 12: 891–897.
Caprari C, Richter A, Bergmann C, Lo Cicero S, Salvi G, Cervone F, De Lorenzo G (1993) Cloning and characterization of a gene encoding the endopolygalacturonase of Fusarium moniliforme. Mycol. Res. 97: 497–505.
Federici F (1985) Production, purification and partial characterization of an endo-polygalacturonase from Cryptococcus albidus var. albidus. Antonie van Leeuwenhoek 51: 139–150.
Gainvors A, Belarbi A (1995) Detection method for polygalacturonase-producing strains of Saccharomyces cerevisiae. Yeast 11: 1493–1499.
Gross KC (1982) A rapid and sensitive spectrophotometric method for assaying polygalacturonase using 2-cyanoacetamide. Hort. Sci. 17: 933–934.
Honda S, Matsuda Y, Takahashi M, Kakehi K, Ganno S (1980) Fluorimetric determination of reducing carbohydrates with 2-cyanoacetamide and application to automated analysis of carbohydrates as borate complexes. Anal. Chem. 52: 1079–1082.
Honda S, Nishimura Y, Takahashi M, Chiba H, Kakehi K (1982) A manual method for the spectrophotometric determination of reducing carbohydrates with 2-cyanoacetamide. Anal. Biochem. 119: 194–199.
Laing E, Pretorius IS (1993) Co-expression of an Erwinia chrysanthemi pectate lyase-encoding gene (pelE) and an E. carotovora polygalacturonase-encoding gene (phehl) in Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 39: 181–188.
Lim J, Yamasaki Y, Suzuki Y, Ozawa J (1980) Multiple forms of endopolygalacturonase from Saccharomyces fragilius. Agric. Biol. Chem. 44: 473–480.
Luh BS, Phaff HJ (1951) Studies on polygalacturonase of certain yeasts. Arch. Biochem. Biophys. 33: 212–227.
Pilar B, Carme S, Tomas GV (1999) Production of pectic enzymes in yeasts. FEMS Microbiol. Lett. 175: 1–9.
Zacharius RM, Zell TE, Morrison JH, Woodlock JJ (1969) Glycoprotein sttaining following electrophoresis on acrylamide gels. Anal. Biochem. 30: 148–152.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miura, T., Abe, F., Inoue, A. et al. Purification and characterization of novel extracellular endopolygalacturonases from a deep-sea yeast, Cryptococcus sp. N6, isolated from the Japan Trench. Biotechnology Letters 23, 1735–1739 (2001). https://doi.org/10.1023/A:1012488115482
Issue Date:
DOI: https://doi.org/10.1023/A:1012488115482