Abstract
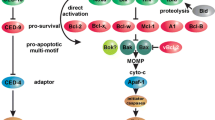
The Bcl-2 family includes a growing number of proteins that play an essential role in regulating apoptosis or programmed cell death. Members of this family display diverse biological functions and can either inhibit or promote cell death signals. Abnormal gene expression of some Bcl-2 family members such as Bcl-2 that inhibits apoptosis is found in a wide variety of human cancers and contributes to the resistance of tumor cells to conventional therapies through interfering with the cell death signals triggered by chemotherapeutic agents. As such, elucidating the structure-function and mechanism of the Bcl-2 family is important for understanding some of the fundamental principles underling the death and survival of cells and of practical value for developing potential therapeutics to control apoptosis in pathological processes. Synthetic peptides derived from homologous or heterogeneous domains in Bcl-2 family proteins that might mediate different biological activities provide simplified and experimentally more tractable models as compared to their full-length counterparts to dissect and analyze the complex functional roles of these proteins. Non-peptidic molecules identified from random screening of natural products or designed by rational structure-based techniques can mimic the effect of synthetic peptides by targeting similar active sites on a Bcl-2 family member protein. In this article, we review recent progress in using these synthetic peptides and non-peptidic mimic molecules to obtain information about the structure and function of Bcl-2 family proteins and discuss their application in modulating and studying intracellular apoptotic signaling.
Similar content being viewed by others
References
Thompson CB. Apoptosis in the pathogenesis and treatment of disease. Science 1995; 267: 1456-1462.
Thornberry N, Lazebnik Y. Caspases: Enemies within. Science 1998; 281: 1312-1316.
Green DR, Reed JC. Mitochondria and apoptosis. Science 1998; 281: 1309-1312.
Ashkenazi A, Dixit VM. Death receptors: Signaling and modulation. Science 1998; 281: 1305-1308.
Adams J, Cory S. The Bcl-2 protein family: Arbiters of cell survival. Science 1998; 281: 1322-1326.
Chao D, Korsmeyer S. Bcl-2 family: Regulators of cell death. Annu Rev Immunol 1998; 16: 395-419.
Petros AM, Medek A, Nettesheim DG, et al. Solution structure of the antiapoptotic protein Bcl-2. Proc Natl Acad Sci USA 2001; 98: 3012-3017.
Muchmore SW, Sattler M, Liang H, et al. X-ray and NMR structure of human Bcl-xL, an inhibitor of programmed cell death. Nature 1996; 381: 335-341.
Aritomi M, Kunishima N, Inohara N, Ishibashi Y, Ohta S, Morikawa K. Crystal structure of rat Bcl-xL. Implications for the function of the Bcl-2 protein family. J Biol Chem 1997; 272: 27886-27892.
Suzuki M, Youle RJ, Tjandra N. Structure of Bax: Coregulation of dimer formation and intracellular localization. Cell 2000; 103: 645-654.
McDonnell JM, Fushman D, Milliman CL, Korsmeyer SJ, Cowburn D. Solution structure of the proapoptotic molecule Bid: A structural basis for apoptotic agonists and antagonists. Cell 1999; 96: 625-634.
Chou JJ, Li H, Salvesen GS, Yuan J, Wagner G. Solution structure of Bid, an intracellular amplifier of apoptotic signaling. Cell 1999; 96: 615-624.
Yang E, Zha J, Jockel J, Boise L, Thompson C, Korsmeyer S. Bad, a heterodimeric partner for Bcl-xL and Bcl-2, displaces Bax and promotes cell death. Cell 1995; 80: 285-291.
Oltvai ZN, Milliman CL, Korsmeyer SJ. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 1993; 74: 609-619.
Reed JC, Miyashita T, Takayama S, et al. Bcl-2 family proteins: Regulators of cell death involved in the pathogenesis of cancer and resistance to therapy. J Cell Biochem 1996; 60: 23-32.
Sattler M, Liang H, Nettesheim D, et al. Structure of BclxL-Bak peptide complex: Recognition between regulators of apoptosis. Science 1997; 275: 983-986.
Desagher S, Osen-Sand A, Nichols A, et al. Bid-induced conformational change of Bax is responsible for mitochondrial cytochrome c release during apoptosis. J Cell Biol 1999; 144: 891-901.
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ. Serine phosphorylation of death agonist Bad in response to survival factor results in binding to 14-3-3 not Bcl-xL. Cell 1996; 87: 619-628.
Slee EA, Keogh SA, Martin SJ. Cleavage of Bid during cytotoxic drug and UV radiation-induced apoptosis occurs downstream of the point of Bcl-2 action and is catalysed by caspase-3: A potential feedback loop for amplification of apoptosisassociated mitochondrial cytochrome c release. Cell Death & Differentiation 2000; 95: 556-565.
Li H, Zhu H, Xu CJ, Yuan J. Cleavage of Bid by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 1998; 94: 491-501.
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X. Bid, a Bcl-2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 1998; 94: 481-490.
Knudson CM, Korsmeyer SJ. Bcl-2 and Bax function independently to regulate cell death. Nat Genet 1997; 16: 358-363.
Minn AJ, Velez P, Schendel SL, et al. Bcl-xL forms an ion channel in synthetic lipid membranes. Nature 1997; 385: 353-357.
Schendel SL, Xie Z, Montal MO, Matsuyama S, Montal M, Reed JC. Channel formation by antiapoptotic protein Bcl-2. Proc Natl Acad Sci USA 1997; 94: 5113-5118.
Schlesinger PH, Gross A, Yin XM, et al. Comparison of the ion channel characteristics of proapoptotic BAX and antiapoptotic Bcl-2. Proc Natl Acad Sci USA 1997; 94: 11357-11362.
Antonsson B, Conti F, Ciavatta A, et al. Inhibition of Bax channel-forming activity by Bcl-2. Science 1997; 277: 370-372.
Schendel SL, Azimov R, Pawlowski K, Godzik A, Kagan BL, Reed JC. Ion channel activity of the BH3 only Bcl-2 family member, BID. J Biol Chem 1999; 274: 21932-21936.
Tsujimoto Y, Shimizu S. Bcl-2 family: Life-or-death switch. FEBS Lett 2000; 466: 6-10.
Shimizu S, Narita M, Tsujimoto Y. Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 1999; 399: 483-487.
Reed JC. Bcl-2 and the regulation of programmed cell death. J Cell Biol 1994; 124: 1-6.
Webb A, Cunningham D, Cotter F, et al. Bcl-2 antisense therapy in patients with non-Hodgkin lymphoma. Lancet 1997; 349: 1137-1141.
Jansen B, Schlagbauer-Wadl H, Brown B, et al. Bcl-2 antisense therapy chemosensitizes human melanoma in SCID mice. Nat Med 1998; 4: 232-234.
Piche A, Grim J, Rancourt C, Gomez-Navarro J, Reed JC, Curiel DT. Modulation of Bcl-2 protein levels by an intracellular anti-Bcl-2 single-chain antibody increases drug-induced cytotoxicity in the breast cancer cell line MCF-7. Cancer Res 1998; 58: 2134-2140.
Kelekar A, Thompson CB. Bcl-2-family proteins: The role of the BH3 domain in apoptosis. Trends Cell Biol 1998; 8: 324-330.
Kelekar A, Chang B, Harlan J, Fesik S, Thompson C. Bad is a BH3 domain-containing protein that forms an inactivating dimer with Bcl-xL. Mol Cell Biol 1997; 17: 7040-7046.
Petros AM, Nettesheim DG, Wang Y, et al. Rationale for BclxL/Bad peptide complex formation from structure, mutagenesis, and biophysical studies. Protein Sci 2000; 9: 2528-2534.
Ottilie S, Diaz JL, Horne W, et al. Dimerization properties of human Bad. Identification of a BH-3 domain and analysis of its binding to mutant Bcl-2 and Bcl-xL proteins. J Biol Chem 1997; 272: 30866-30872.
Diaz JL, Oltersdorf T, HorneW, et al. A common binding site mediates heterodimerization and homodimerization of Bcl-2 family members. J Biol Chem 1997; 272: 11350-11355.
Cosulich S, Worrall V, Hedge P, Green S, Clarke P. Regulation of apoptosis by BH3 domains in a cell-free system. Curr Biol 1997; 7: 913-920.
Jurgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC. Bax directly induces release of cytochrome c from isolated mitochondria. Proc Natl Acad Sci USA 1998; 95: 4997-5002.
Narita M, Shimizu S, Ito T, et al. Bax interacts with the permeability transition pore to induce permeability transition and cytochrome c release in isolated mitochondria. Proc Natl Acad Sci USA 1998; 95: 14681-146816.
Morgan BA, Finnegan NM, Prevost G, Cotter TG. 91st Annual Meeting of the American Association for Cancer Research 2000; 42: 4693.
Shimizu S, Konishi A, Kodama T, TsujimotoY. BH4 domain of antiapoptotic Bcl-2 family members closes voltage-dependent anion channel and inhibits apoptotic mitochondrial changes and cell death. Proc Natl Acad Sci USA 2000; 97: 3100-3105.
Huang DC, Adams JM, Cory S. The conserved N-terminal BH4 domain of Bcl-2 homologues is essential for inhibition of apoptosis and interaction with CED-4. EMBO J 1998; 17: 1029-1039.
Theodore L, Derossi D, Chassaing G, et al. Intraneuronal delivery of protein kinase C pseudosubstrate leads to growth cone collapse. J Neurosci 1995; 15: 7158-7167.
Ball KL, Lain S, Fahraeus R, Smythe C, Lane DP. Cell-cycle arrest and inhibition of Cdk4 activity by small peptides based on the carboxy-terminal domain of p21WAF1. Curr Biol 1997; 7: 71-80.
Bonfanti M, Taverna S, Salmona M, D'Incalci M, Broggini M. p21WAF1-derived peptides linked to an internalization peptide inhibit human cancer cell growth. Cancer Res 1997; 57: 1442-1446.
Fahraeus R, Paramio JM, Ball KL, Lain S, Lane DP. Inhibition of pRb phosphorylation and cell-cycle progression by a 20-residue peptide derived from p16CDKN2/INK4A. Curr Biol 1996; 6: 84-91.
Selivanova G, Iotsova V, Okan I, et al. Restoration of the growth suppression function of mutant p53 by a synthetic peptide derived from the p53 C-terminal domain. Nat Med 1997; 3: 632-638.
Williams EJ, Dunican DJ, Green PJ, et al. Selective inhibition of growth factor-stimulated mitogenesis by a cell-permeable Grb2-binding peptide. J Biol Chem 1997; 272: 22349-22354.
Hall H, Williams EJ, Moore SE, Walsh FS, Prochiantz A, Doherty P. Inhibition of FGF-stimulated phosphatidylinositol hydrolysis and neurite outgrowth by a cell-membrane permeable phosphopeptide. Curr Biol 1996; 6: 580-587.
Holinger EP, Chittenden T, Lutz RJ. Bak BH3 peptides antagonize Bcl-xL function and induce apoptosis through cytochrome c-independent activation of caspases. J Biol Chem 1999; 274: 13298-13304.
Ioannides CG, Freedman RS, Liskamp RM, Ward NE, O'Brian CA. Inhibition of IL-2 receptor induction and IL-2 production in the human leukemic cell line Jurkat by a novel peptide inhibitor of protein kinase C. Cell Immunol 1990; 131: 242-252.
Kole HK, Garant MJ, Kole S, Bernier M. A peptide-based protein-tyrosine phosphatase inhibitor specifically enhances insulin receptor function in intact cells. J Biol Chem 1996; 271: 14302-14307.
Wang JL, Zhang ZJ, Choksi S, et al. Cell permeable Bcl-2 binding peptides: A chemical approach to apotosis induction in tumor cells. Cancer Res 2000; 60: 1498-1502.
Green M, Loewenstein PM. Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell 1988; 55: 1179-1188.
Derossi D, Calvet S, Trembleau A, Brunissen A, Chassaing G, Prochiantz A. Cell internalization of the third helix of the Antennapedia homeodomain is receptor-independent. J Biol Chem 1996; 271: 18188-18193.
Schwarze SR, Ho A, Vocero-Akbani A, Dowdy SF. In vivo protein transduction: Delivery of a biologically active protein into the mouse. Science 1999; 285: 1569-1572.
Tomita F, Tamaoki T, Shirahata K, et al. Novel antitumor antibiotics, tetrocarcins. J Antibiot (Tokyo) 1980; 33: 668-670.
Nakashima T, Miura M, Hara M. Tetrocarcin A inhibits mitochondrial functions of Bcl-2 and suppresses its anti-apoptotic activity. Cancer Research 2000; 60(5): 1229-1235.
Tzung SP, Kim KM, Basanez G, Zimmerberg J, Zhang KY, Hockenbery DM. Antimycin A mimics a BH3 domaincontaining peptide and selectively induces apoptosis in cell lines overexpressing Bcl-xL. Proc AACR-NCI-EORTIC International Conference, Molecular Target and Cancer Therapeutics: Discovery, Development and Clinical Validation.Washington DC, USA 1999; 87-88.
Strong FM. Isolation, structure and properties of antimycin A. Topics in Microbial Chemistry. NY. USA: John Wiley & Sons Inc, 1956: 1-43.
Tokutake N, Miyoshi H, Satoh T, Hatano T, Iwamura H. Structural factors of antimycin A molecule required for inhibitory action. Biochim Biophys Acta 1994; 1185: 271-278.
Yin XM, Oltvai ZN, Korsmeyer SJ. BH1 and BH2 domains of Bcl-2 are required for inhibition of apoptosis and heterodimerization with Bax. Nature 1994; 369: 321-323.
Chittenden T, Flemington C, Houghton AB, et al. A conserved domain in Bak, distinct from BH1 and BH2, mediates cell death and protein binding functions. EMBO J 1995; 14: 5589-5596.
Boyd JM, Gallo GJ, Elangovan B, et al. Bik, a novel deathinducing protein shares a distinct sequence motif with Bcl-2 family proteins and interacts with viral and cellular survival-promoting proteins. Oncogene 1995; 11: 1921-1928.
Zha H, Aime-Sempe C, Sato T, Reed JC. Proapoptotic protein Bax heterodimerizes with Bcl-2 and homodimerizes with Bax via a novel domain (BH3) distinct from BH1 and BH2. J Biol Chem 1996; 271: 7440-7444.
Huang Z. Structural chemistry and therapeutic intervention of protein-protein interaction in immune response, HIV entry and apoptosis. Pharmacology & Therapeutics 2000; 86: 201-215.
Wang J, Liu D, Zhang Z, et al. Structure-based discovery of a novel organic compound that binds Bcl-2 protein and induces apoptosis of tumor cells. Proc Natl Acad Sci USA 2000; 97: 7124-7129.
Yu N, Aramini JM, Germann MW, Huang Z. Reactions of salicylaldehydes with alkyl cyanoacetates on the surface of solid catalysts: Syntheses of 4H-chromene derivatives. Tetrahedron Lett 2000; 41:6993-6996.
Sedlak TW, Oltvai ZN, Yang E, et al. Multiple Bcl-2 family members demonstrate selective dimerizations with Bax. Proc Natl Acad Sci USA 1995; 92: 7834-7838.
Shibasaki F, Kondo E, Akagi T, McKeon F. Suppression of signalling through transcription factor NF-AT by interactions between calcineurin and Bcl-2. Nature 1997; 386: 728-731.
Wang HG, Rapp UR, Reed JC. Bcl-2 targets the protein kinase Raf-1 to mitochondria. Cell 1996; 87: 629-638.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, D., Huang, Z. Synthetic peptides and non-peptidic molecules as probes of structure and function of Bcl-2 family proteins and modulators of apoptosis. Apoptosis 6, 453–462 (2001). https://doi.org/10.1023/A:1012481406064
Issue Date:
DOI: https://doi.org/10.1023/A:1012481406064