Abstract
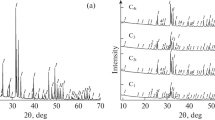
Hydroxyapatite (HAP) and tri-calcium phosphate (TCP) powders and coatings with a Ca/P molar ratio from 1.56 to 1.77 were prepared by the sol-gel technique using calcium 2-ethylhexanoate (Ca(O2C8H15)2) and 2-ethyl-hexyl-phosphate as calcium and phosphorus precursors, respectively. The structural evolution and phase formation mechanisms of HAP and tri-calcium phosphate in calcined powders and coatings on Si wafer and Ti-alloy substrates (Ti-30Nb-3Al and Ti-5Al-2.5Fe) were characterized by X-ray diffraction, Fourier transform infrared spectroscopy (FTIR) and scanning electron microscopy (SEM). The elimination of organics was studied by differential thermal analysis (DTA) and thermogravimetry (TGA). Two different formation mechanisms of crystallization are proposed. In sols with Ca/P ≤ 1.67, β-tricalcium phosphate is formed as the major phase and hydroxyapatite as a minor phase by calcination at 700°C. At 900°C these phases react to form AB-type carbonated hydroxyapatite (Ca10−2x/3[(PO4)6−x (CO3) x ][(OH)2−x/3−2y (CO3) y ]). A release of CO2 substituting PO4 3− occurs between 900°C and 1100°C yielding carbonate apatite, Ca10(PO4)6[(OH)2−2y (CO3) y ], whereas CO2 substituting OH− groups in the apatite structure is released above 1200°C. In sols with Ca/P ≥ 1.70, rather than carbonate apatite, B-carbonated hydroxyapatite Ca10−2x/3[(PO4)6−x (CO3) x ](OH)2 is formed, which subsequently decomposes into HAP and CaO above 1200°C. The optimum sintering conditions for coatings on Ti-alloys are found to be 600°C for 10 minutes, since, at higher temperature, oxidation of titanium and the formation of rutile (TiO2) occur. Dip coating and sintering in two cycles yielded a homogeneous and dense coated film with a thickness of 250 nm.
Similar content being viewed by others
References
R. J. Furlong and J. F. Osborn, J. Bone Joint Surg. 5 (1991) 741.
R. Evasic, Dentistry Todays 2 (1993) 90.
P. Ducheyne, S. Radin, M. Heughebaert and J. C. Heughebaert, Biomaterials 11 (1990) 244.
D. M. Liu, H. M. Chou and J. D. Wu, J. Mater. Sci.: Mater. Med. 5 (1994) 147.
L. Z. LeGeros, J. P. LeGeros, R. Kijkowska, R. Zhenf, C. Baptista and J. L. Wong, in “Ceramic Transactions, Vol. 48. Bioceramics: Materials and Applications,” edited by G. Fischman, A. Clare and L. Hench (American Ceramic Society, Westerville, OH, 1995) p. 173.
R. D. Bloebaum, D. A. Beeks, L. D. Dorr, J. A. Dupont, C. G. Savory and A. A. Hofman, Proceedings of the 20th Annual Meeting of the Society for Biomaterials, Boston, MA, 1994 (Society for Biomaterials, Mineapolis, MN, 1994) p. 125.
L. Torissi and G. Foti, Appl. Phys. Lett. 62 (1993) 237.
C. M. Cotell, Appl. Surf. Sci. 69 (1993) 140.
Y. Fujishiro, T. Sato and A. Okuwaki, J. Mater. Sci.: Mater. Med. 6 (1995) 172.
M. Tanachashi, T. Yao, T. Kokubo, M. Minoda, T. Nakamura and T. Yamamura, ibid. 6 (1995) 319.
T. Kokubo, K. Hata, T. Nakamura and T. Yamamuro, in “Bioceramics,” Vol. 4 edited by W. Bonfield, G. W. Hasting and K. E. Tanner (Butherworth Heinemann Ltd., 1991) p. 113.
H. B. Wen, J. R. De Wijn, Q. Liu, K. De Groot and F. Z. Cui, J. Mater. Sci.: Mater. Med. 8 (1997) 765.
C. J. Brinker and G.W. Scherrer, “Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing” (Academic Press, New York, 1992) p. 845.
A. Deptula, J. Rebandel, W. Drozda, W. Lada and T. Olczak, in “Better Ceramics Through Chemistry,” edited by V. M. J. Hampden-Smith, W. G. Klemperer and C. J. Brinnker (Mat. Res. Soc. Symp. Proc. 270, Pittsburgh, PA, 1992) p. 277.
Y. Masuda, K. Matubaraand and S. Sakka, J. Ceram. Sco. Jpn. 98 (1990) 84.
T. Brendel, A. Engel and C. Ruessel, J. Mater. Sci.: Mater. Med. 3 (1992) 175.
Q. Quii, P. Vincent, B. Lowenberg, M. Sayer and J. E. Davies, Cells Mater. 3 (1993) 351.
W. Weng and J. P. Baptista, J. Am. Ceram. Soc. 82 (1999) 27.
C. M. Lopatin, T. L. Alford, V. B. Pizziconi and T. Laursen, Mater. Letters 37 (1998) 211.
S. W. Russell, K. A. Luptak, C. T. A. Suchicital, T. L. Alford and V. B. Pizziconi, J. Am. Ceram. Soc. 79 (1996) 837.
M. Sauer, R. Nonninger and H. Schmidt, in Werkstoffwoche' 98, Werstoffe für Medizintechnik Band IV Symp. 4, editors H. Planck and H. Stallforth (Wiley-VCH, Weinheim, New York, 1999) p. 67.
M. Hosonuma and T. Shimamune, “Process for producing metal composite materials having a coating of calcium phosphate for bone and tooth implants,” Patent EP 310574 A1 19890405 EP 88-830391 19880928.
I. C. Ison, M. T. Fulmer, B. M. Barr and B. R. Constantz, in “Hydroxyapatite and related materials,” editors P. W. Brown and B. Constantz (CRC Press, 1994) p. 215.
I. Rehman and W. Bonfield, J. Mater. Sci.: Mater. Med. 8 (1997) 1.
P. Layrolle and A. Lebugle, Chem. Mater. 6 (1994) 1996.
B. Narasaraju and D. E. Phebe, J. Mater. Sci. 31 (1996) 1.
Suetsugu, I. Shimoya and J. Tanaka, J. Am. Ceram. Soc. 81 (1998) 746.
R. Rao, H. N. Roopa and T. S. Kannan, J. Mater. Sci.: Mater. Med. 8 (1997) 511.
P. Layrolle, A. Ito and T. Tateishi, J. Am. Ceram. Soc. 81 (1998) 1421.
A. Tampieri, G. Celotti, F. Szontagh and E. Landi, J. Mater. Sci.: Mater. Med. 8 (1997) 29.
E. Park and R. A. Condrate Sr., Mater. Letters 40 (1999) 228.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tkalcec, E., Sauer, M., Nonninger, R. et al. Sol-gel-derived hydroxyapatite powders and coatings. Journal of Materials Science 36, 5253–5263 (2001). https://doi.org/10.1023/A:1012462332440
Issue Date:
DOI: https://doi.org/10.1023/A:1012462332440