Abstract
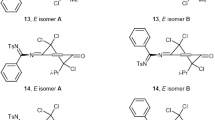
The chlorination and bromination of 2,3-dimethyl-, 3-methyl-6-isopropyl-, and 2,6-diisopropyl-4-aroyl(or arylsulfonyl)oxyimino-2,5-cyclohexadienones follow the proposed rules of halogenation of 4-aroyl(or arylsulfonyl)oxyimino-2,5-cyclohexadienones: the reaction occurs preferentially at the cis-CÍC bond of the quinoid ring; simultaneous halogenation at both double bonds is not observed; halogen adds mainly across unsubstituted CÍC bond; no halogenation occurs at the double bond already substituted by a halogen; bromination of the CÍC bond with an alkyl substituent is more difficult than chlorination; the second halogen molecule adds only after regioselective dehydrohalogenation.
Similar content being viewed by others
REFERENCES
Avdeenko, A.P. and Glinyanaya, N.M., Russ. J. Org. Chem., 1995, vol. 31, no. 11, pp. 1507-1513.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, V.V., Russ. J. Org. Chem., 1996, vol. 32, no. 1, pp. 85-89.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, V.V., Russ. J. Org. Chem., 1995, vol. 31, no. 10, pp. 1380-1385.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, V.V., Zh. Org. Khim., 1993, vol. 29, no. 7, pp. 1402-1411.
Baldwin, J.E. and Norris, R.K., J. Org. Chem., 1981, vol. 46, no. 6, pp. 697-703.
Perrin, C.L. and Engler, R.E., J. Org. Chem., 1997, vol. 62, no. 3, pp. 687-692.
Titov, E.A. and Burmistrov, S.I., Ukr. Khim. Zh., 1960, vol. 26, no. 6, pp. 744-749.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Avdeenko, A.P., Zhukova, S.A. & Konovalova, S.A. Halogenation of Some Alkyl-Substituted 4-Aroyloxyimino- and 4-Arylsulfonyloxyimino-2,5-cyclohexadienones. Russian Journal of Organic Chemistry 37, 382–387 (2001). https://doi.org/10.1023/A:1012403708255
Issue Date:
DOI: https://doi.org/10.1023/A:1012403708255