Abstract
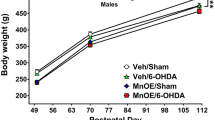
In the present study the effects of chronic manganese (Mn) treatment on adenosine A2a receptor binding in mouse brain have been assessed. Male albino mice were divided in two groups: In the Mn-treated group, the animals were injected intraperitoneally (i.p.) with MnCl2 (5 mg/kg/day) five days per week during 9 weeks; in the control group, they were injected likewise with a saline solution. A significant decrease of the Kd without alteration of Bmax in the cerebellum and, an increase of the Kd and Bmax in hippocampus of mice treated with Mn were found. Also, an increase of Kd in frontal cortex was observed. The binding parameters in caudate nucleus, olfactory bulb and hypothalamus were not altered by Mn. A significant decrease in the adenosine concentration in caudate nucleus, olfactory bulb and hypothalamus, without significant changes in hippocampus, frontal cortex and cerebellum was also detected. These findings suggest that chronic administration of Mn could affect adenosine receptor function and turnover, depending on the brain region analyzed.
Similar content being viewed by others
REFERENCES
Huang, C. C., Chu, N. S., Lu, C. S., Wang, J. D., Tsai, J. L., Tzeng, J. L., Walters, E. C., and Calne, D. B. 1989. Chronic manganese intoxication. Arch. Neurol. 46:1104–1106.
Bonilla, E. 1987. El manganeso y su importancia biomédica. Ediciones Astro Data Maracaibo, Venezuela. pp. 2–79.
Doherty, J. D., Lourte, C. J., and Trams, E. G. 1983. Mn2+ stimulated ATPase in rat brain. Neurochem. Res. 8:493–500.
Cooper, D. M. F. and Caldwell, K. K. 1990. Signal transduction mechanisms for adenosine. In: Adenosine and adenosine receptors. Williams M. (Eds), The Humana Press, Clifton, New Jersey, pp. 105–141.
Bonilla, E. 1981. Manganismo crónico experimental: Aspectos neuroquímicos. Invest. Clin. 22:159–173.
Mena, I., Marín, O., Fuenzalida, S., and Cotzias, G. C. 1967. Chronic manganese poisoning. Clinical picture and manganese turnover. Neurology 17:128–136.
Bhargava, H. N. 1987. Effect of repeated administration of manganese on striatal cholinergic and dopaminergic receptors in the rat. Toxicol. Lett. 37:135–141.
Chandra, S. V. and Shukla, G. 1981. Effect of manganese on synthesis of brain catecholamines in growing rats. Acta Pharmacol. Toxicol. 48:349–354.
Bonilla, E. and Diez-Ewald, M. 1974. Effect of L-dopa on brain concentration of dopamine and homovanillic acid in rats after chronic manganese chloride administration. J. Neurochem. 22:297–299.
Barbeau, A. 1984. Manganese and extrapyramidal disorders. Neurotoxicol. 5:13–36.
Cano, G., Bonilla, E., Castro, F., Alburges, M., and Wamsley, J. 1992. Intoxicación crónica con manganeso. Cuantificación autorradiográfica de los receptores colinérgicos muscarínicos en el cerebro de ratón. Invest. Clin. 33:69–80.
Villalobos, V., Bonilla, E., Estévez, J., Dávila, J. O., and Castro, F. 1994. Manganese toxicity: Muscarinic receptor binding in the mouse brain. J. Toxicol. Environ. Health 42:185–191.
Díaz, H. L. and Gómez, G. A. 1993. Neurobiología de la intoxicación por manganeso. Rev. Chil. Neuro-Psiquiat. 31:351–358.
Coassin, M., Ursini, F., and Bindoli, A. 1992. Antioxidant effect of manganese Arch. Biochem. Biophys. 299:330–333.
Pugh, S. Y., Guiseppi, J. L., and Fridovich, I. 1984. Induction of superoxide dismutase in Escherichia coli by manganese and iron. J. Bacteriol. 160:137–142.
Shiffmann, S. N., Jacobs, O. P., and Vanderhaeghen, J. J. 1991. The striatal restricted adenosine A2 receptor (RDC) is expressed by enkephalin but not by substance P neurons. An in situ hibridization histochemistry study. J. Neurochem. 57:1062–1067.
Wojcik W. J. and Neff, N. H. 1983. Differential location of adenosine A1 and A2 receptors in striatum. Neurosci. Lett. 41:55–60.
Jarvis, M. F. and Williams, M. 1990. Adenosine in Central Nervous System. In: Adenosine and adenosine receptors. Williams M. (Eds), The Humana Press, Clifton, New Jersey, pp. 423–474.
Jarvis., M. F. and Williams, M. 1989. Direct autoradiographic localization of adenosine A2 receptors in the rat brain using the selective A2-selective agonist, [3H]CGS 21680. Eur. J. Pharmacol. 168:243–246.
Jarvis, M. F., Schulz, R., Hutchison, A. J., Do, U. H., Sills, M. A., and Williams, M. 1989. [3H]CGS 21680, a selective A2 adenosine receptor agonist, directly labels A2 receptors in rat brain. J. Pharmacol. Exp. Ther. 251:888–893.
Johansson, B., Parkinson, F. E., and Fredholm, B. 1972. Effects of mono-and divalent ions on the binding of the adenosine analogue CGS 21680 to adenosine A2 receptors in rat striatum. Biochem. Pharmacol. 44:2365–2370.
Latini, S., Pazzagli, M., Pepeu, G., and Pedata, F. 1996. A2 adenosine receptors. Their presence and neuromodulatory role in the central nervous system. Gen. Pharmacol. 27:925–933.
Fink, J. K., Weaver, D. R., Rivkess, S. A., Peterfrenund, R. A., Pollack, A., Adler, E. M., and Reppert, S. M. 1992. Molecular cloning of the rat A2 adenosine receptor: Selective coexpression with D2 dopamine receptors in rat striatum. Mol. Res. 14:186–195.
Albin, R. L. Young, A. B., and Penney, B. 1989. The functional anatomy of basal ganglia disorders. Trends Neurosci. 12:366-375.
Glowisnski, J. and Iversen, L. 1966. Regional studies of catecholamines in the rat brain. I. The disposition of 3H-norepinephrine, 3H-dopamine and 3H-DOPA in various regions of the brain. J. Neurochem. 13:655–669.
Bonilla, E., Arrieta, A., Castro, F., Dávila, J. O., and Quiroz, I. 1994. Manganese toxicity: Free aminoacids in striatum and olfactory bulb of the mouse. Invest. Clin. 35:175–181.
Lowry, O. H., Rosebrouh, N. J., Farr, A. L., and Randall, R. L. 1951. Protein measurement with folin phenol reagent. J. Biol. Chem. 193:265–275.
Bonilla, E. 1978. Flameless atomic absorption spectrophotometric determination of manganese in rat brain and other tissues. Clin. Chem. 24:471–474.
Munson, P. J. and Rodbar, D. 1980. Ligand. A versatile computarized approach for characterization of ligand bindingsystems. Anal. Biochem. 107:220–239.
Betto, P., Popoli, P., Ricciarello, G., Coporali, M. G., and Antonini, R. 1994. Simultaneous high-performance liquid chromatographic determination of adenosine and dopamine in rat striatal tissue with combined ultraviolet absorbance and electrochemical detection. J. Cromatogr. B. 662:21–25.
Turbochrom 4 Tutorial 1995. S270-0604-C PE Nelson Division. Perkin Elmer, USA.
Barbeau, A. 1984. Manganese and extrapyramidal disorders. Neurotoxicol. 5:13–36.
Cardozo, J. and Bonilla, E. 1985. The neuropathology of experimental chronic manganese poisoning in rats. A preliminary report. Invest. Clin. 26:117–124.
Donaldson, J. 1987. The physiopathologic significance of manganese in brain. Its relation to schizophrenia and neurodegenerative disorders. Neurotoxicol. 8:451–462.
Gerfen, C. R., Engber, T. M., Mahan, L. C., Susel, Z., Chase, T. N., Monsma, F. J., and Sibley D. R. 1990. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 250:1429–1432.
Le Moine, C., Normand, E., Guitteny, A. F., Fouque, B., Teoule, R., and Bloch, B. 1990. Dopamine receptor gene expression by enkephalin neurons in rat forebrain. Proc. Natl. Acad. Sci. USA 87:230–234.
Gerfen, C. R. 1992. The neostriatal mosaic: Multiple levels of compartmental organization. Trends Neurosci. 15:133–139.
Pedata, F., Pazzagli, M., Tilii, S., and Pepeu, G. 1990. Regional differences in the electrically stimulated release of endogenous and radioactive adenosine and purine derivatives from rat brain slices. Naunyn. Schmiedeberg's Arch. Pharmacol. 342:447–453.
Ferré, S., Herrera-Marschitz, M., Grabowska-Ande, M., Ungerstedt, U., Casas, M., and Anden, N. E. 1991. Postsynaptic dopamine/adenosine interaction: Adenosine analogues inhibit a D2 mediated behaviour in short-term reserpined mice. Eur. J. Pharmacol. 192:30–35.
Ferré, S., Herrera-Marschitzz, M., Grabowska-Ande, M., Ungerstedt, U., Casas, M., and Anden, N. E. 1991. Postsynaptic dopamine/adenosine interaction: II Postsynaptic dopamine agonism and adenosine antagonism of methylxantines in short-term reserpined mice. Eur. J. Pharmacol. 192:36–42.
Richardson, P. J., Kase, H., and Jenner, P. G. 1980. Adenosine A2a receptor antagonists as new agents for the treatment of Parkinson's disease. Trends Pharmacol. Sci. 18:338–344.
Bonilla, E. 1980. L-Tyrosine hydroxylase activity in the rat brain after chronic oral administration of manganese chloride. Neurobehav. Toxicol. 2:37–41.
Cotzias, G. C., Papavasiliou, P. S., Mena, I., Tang, L. C., and Miller, S. T. 1974. Manganese and catecholamines. Adv. Neurol. 5:235–243.
Ferré, S., Rubio, A., and Fuxe, K. 1991. Stimulation of adenosine A2 receptors induces catalepsy. Neurosci. Lett. 130:162–164.
Johansson, B., Georgiev, V., Parkinson, F. E., and Fredholm, B. B. 1993. The binding of the adenosine A2 receptor selective agonist [3H]CGS 21680 to rat cortex differs from its binding to rat striatum. Eur. J. Pharmacol. 247:103–110.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Villalobos, V., Suárez, J., Estévez, J. et al. Effect of Chronic Manganese Treatment on Adenosine Tissue Levels and Adenosine A2a Receptor Binding in Diverse Regions of Mouse Brain. Neurochem Res 26, 1157–1161 (2001). https://doi.org/10.1023/A:1012379024571
Issue Date:
DOI: https://doi.org/10.1023/A:1012379024571