Abstract
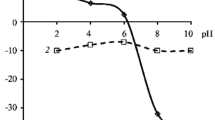
The electrophoretic mobility of particles of silicon dioxide, aluminum hydroxide, and iron hydroxide was measured as a function of pH in NaCl and KCl background solutions. Isoelectric points for the investigated objects were determined. Electrokinetic potentials were calculated with allowances for the particle shape and the polarization of the electrical double layer within the framework of the Overbeek–Boes–Wiersema model.
Similar content being viewed by others
REFERENCES
Klebanov, A.V., Bogdanova, N.F., Ermakova, L.E., et al., Kolloidn. Zh., 2001, vol. 63, no. 5, p. 617.
James, A.M., Surface and Colloid Science, Matijevi, E., Ed., New York: Plenum, 1979, vol. 11, p. 121.
Wiersema, P.H., Loeb, A.L., and Overbeek, J.T.G., J. Colloid Interface Sci., 1966, vol. 22, nos. 1-2, p. 78.
Sidorova, M.P., Zastrow, H., Ermakova, L.E., et al., Kolloidn. Zh., 1999, vol. 61, no. 1, p. 113.
Lyklema, J., Fundamentals of Interface and Colloid Science, vol. II: Solid-Liquid Interfaces, New York: Academic, 1995.
Dukhin, S.S. and Derjaguin, B.V., Elektroforez (Electrophoresis), Moscow: Nauka, 1976.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klebanov, A.V., Bogdanova, N.F., Ermakova, L.E. et al. Electrosurface Properties of Hydr(oxides) and Oxide Nanostructures in 1 : 1 Electrolyte Solutions: 2. Electrokinetic Characteristics of Boehmite, Goethite, and Silicon Dioxide. Colloid Journal 63, 568–572 (2001). https://doi.org/10.1023/A:1012342801199
Issue Date:
DOI: https://doi.org/10.1023/A:1012342801199