Abstract
Purpose. Calculation of the ideal solubility of a crystalline solute in a liquid solvent requires knowledge of the difference in the molar heat capacity at constant pressure of the solid and the supercooled liquid forms of the solute, ΔCp. Since this parameter is not usually known, two assumptions have been used to simplify the expression. The first is that ΔCp can be considered equal to zero; the alternate assumption is that the molar entropy of fusion, ΔSf, is an estimate of ΔCp. Reports claiming the superiority of one assumption over the other, on the basis of calculations done using experimentally determined parameters, have appeared in the literature. The validity of the assumptions in predicting the ideal solubility of five structurally unrelated compounds of pharmaceutical interest, with melting points in the range 420 to 470K, was evaluated in this study.
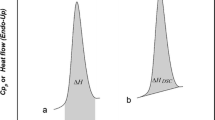
Methods. Solid and liquid heat capacities of each compound near its melting point were determined using differential scanning calorimetry. Linear equations describing the heat capacities were extrapolated to the melting point to generate the differential molar heat capacity.
Results. Linear data were obtained for both crystal and liquid heat capacities of sample and test compounds. For each sample, ideal solubility at 298K was calculated and compared to the two estimates generated using literature equations based on the differential molar heat capacity assumptions.
Conclusions. For the compounds studied, ΔCp was not negligible and was closer to ΔSf than to zero. However, neither of the two assumptions was valid for accurately estimating the ideal solubility as given by the full equation.
Similar content being viewed by others
REFERENCES
S. H. Neau, G. L. Flynn, and S. H. Yalkowsky. Int. J. Pharm. 49:223–229 (1989).
S. H. Yalkowsky, G. L. Flynn, and T. G. Slunick. J. Pharm. Sci. 61:852–857 (1972).
S. H. Yalkowsky, R. J. Orr, and S. C. Valvani. Ind. Eng. Chem. Fund. 18:351–353 (1979).
S. H. Yalkowsky and S. C. Valvani. J. Chem. Eng. Data 24:127–129 (1979).
S. H. Yalkowsky and S. C. Valvani. J. Pharm. Sci. 69:912–922 (1980).
T. A. Hagen and G. L. Flynn. J. Pharm. Sci. 72:409–414 (1983).
S. H. Yalkowsky. J. Pharm. Sci. 70:971–973 (1981).
J. H. Hildebrand, J. M. Prausnitz and R. L. Scott. Regular and Related Solutions, Van Nostrand Reinhold Co., New York, 1970.
J. W. Mauger, A. N. Paruta, and R. J. Gerraughty. J. Pharm. Sci. 61:94–97 (1972).
A. Martin, J. Newburger, and A. Adjei. J. Pharm. Sci. 69:487–491 (1980).
P. J. Richardson, D. F. McCafferty, and A. D. Woolfson. Int. J. Pharm. 78:189–198 (1992).
C. W. S. Subrahmanyam, M. Sreenivasa Reddy, J. Venkata Rao, and P. Gundu Rao. Int. J. Pharm. 78:17–24 (1992).
D. J. W. Grant, M. Mendizadeh, A. H.-L. Chow and J. E. Fairbrother. Int. J. Pharm. 18:25–38 (1984).
S. H. Neau and G. L. Flynn. Pharm. Res. 7:1157–1162 (1990).
D. Mishra and S. H. Yalkowsky. Pharm. Res. 9:958–960 (1992).
M. J. O'Neill. Anal. Chem. 38:1331–1336 (1966).
D. C. Ginnings and G. T. Furukawa. J. Am. Chem. Soc. 75:522–527 (1953).
P. Goursot, H. L. Girdhar, and E. F. Westrum. J. Phys. Chem. 74:2538–2541 (1970).
R. D. Chirico, S. E. Knipmeyer, A. Nguyen, and W. V. Steele. J. Chem. Thermodynamics. 21:1307–1331 (1989).
C. M. L. Atkinson and M. J. Richardson. Trans. Faraday Soc. 65:1764–1773 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Neau, S.H., Bhandarkar, S.V. & Hellmuth, E.W. Differential Molar Heat Capacities to Test Ideal Solubility Estimations. Pharm Res 14, 601–605 (1997). https://doi.org/10.1023/A:1012148910975
Issue Date:
DOI: https://doi.org/10.1023/A:1012148910975