Abstract
Purpose. To define the tubular localization and tissue distribution of PEPT1 (low-affinity, high-capacity transporter) and PEPT2 (high-affinity, low-capacity transporter) in rat kidney.
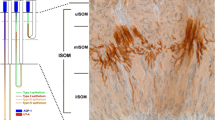
Methods. mRNA expression of PEPT1 and PEPT2 was assessed with reverse transcription-polymerase chain reaction (RT-PCR) methods using cDNA prepared from microdissected nephron segments in rat. Tissue localization of rat renal PEPT1 and PEPT2 mRNA was further assessed by in situ hybridization with radiolabeled probes.
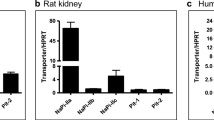
Results. RT-PCR analysis of microdissected segments from rat nephron showed that both PEPT1 and PEPT2 are confined to the proximal tubule. While PEPT1 is specific for early regions of the proximal tubule (pars convoluta), PEPT2 is overwhelmingly but not exclusively expressed in latter regions of the proximal tubule (pars recta). All other segments along the nephron were negative for PEPT1 or PEPT2 mRNA transcripts. These findings were supported by in situ hybridization results in which PEPT1 was selectively expressed in kidney cortex and PEPT2 in the outer stripe of outer medulla.
Conclusions. Contrary to current opinion, the data suggest that peptides are handled in a sequential manner in proximal regions of the nephron, first by the low-affinity, high-capacity transport system and second by the high-affinity, low-capacity transport system.
Similar content being viewed by others
REFERENCES
H. Daniel. Function and molecular structure of brush border membrane peptide/H+symporters. J. Membrane Biol. 154:197-203 (1996).
Y.-J. Fei, Y. Kanai, S. Nussberger, V. Ganapathy, F. H. Leibach, M. F. Romero, S. K. Singh, W. F. Boron, and M. A. Hediger. Expression cloning of a mammalian proton-coupled oligopeptide transporter. Nature 368:563-566 (1994).
M. Boll, M. Herget, M. Wagener, W. M. Weber, D. Markovich, J. Biber, W. Clauss, H. Murer, and H. Daniel. Expression cloning and functional characterization of the kidney cortex high-affinity proton-coupled peptide transporter. Proc. Natl. Acad. Sci. 93:284-289 (1996).
R. Liang, Y.-J. Fei, P. D. Prasad, S. Ramamoorthy, H. Han, T. L. Yang-Feng, M. A. Hediger, V. Ganapathy, and F. H. Leibach. Human intestinal H+/peptide cotransporter: Cloning, functional expression, and chromosomal localization. J. Biol. Chem. 270:6456-6463 (1995).
W. Liu, R. Liang, S. Ramamoorthy, Y.-J. Fei, M. E. Ganapathy, M. A. Hediger, V. Ganapathy, and F. H. Leibach. Molecular cloning of PEPT 2, a new member of the H+/peptide cotransporter family, from human kidney. Biochim. Biophys. Acta 1235:461-466 (1995).
K.-I. Miyamoto, T. Shiraga, K. Morita, H. Yamamoto, H. Haga, Y. Taketani, I. Tamai, Y. Sai, A. Tsuji, and E. Takeda. Sequence, tissue distribution and developmental changes in rat intestinal oligopeptide transporter. Biochim. Biophys. Acta 1305:34-38 (1996).
H. Saito, M. Okuda, T. Terada, S. Sasaki, and K.-I. Inui. Cloning and characterization of a rat H+/peptide cotransporter mediating absorption of β-lactam antibiotics in intestine and kidney. J. Pharmacol. Exp. Ther. 275:1631-1637 (1995).
H. Saito, T. Terada, M. Okuda, S. Sasaki, and K.-I. Inui. Molecular cloning and tissue distribution of rat peptide transporter PEPT2. Biochim. Biophys. Acta 1280:173-177 (1996).
T. C. Freeman, B. S. Bentsen, D. T. Thwaites, and N. L. Simmons. H+/Di-tripeptide transporter (PepT1) expression in the rabbit intestine. Pflügers Arch—Eur. J. Physiol. 430:394-400 (1995).
H. Ogihara, H. Saito, B.-C. Shin, T. Terada, S. Takenoshita, Y. Nagamachi, K.-I. Inui, and K. Takata. Immuno-localization of H+/peptide cotransporter in rat digestive tract. Biochem. Biophys. Res. Commun. 220:848-852 (1996).
M. Brandsch, C. Brandsch, P. D. Prasad, V. Ganapathy, U. Hopfer, and F. H. Leibach. Identification of a renal cell line that constitutively expresses the kidney-specific high-affinity H+/peptide cotransporter. FASEB J. 9:1489-1496 (1995).
F. H. Leibach and V. Ganapathy. Peptide transporters in the intestine and the kidney. Annu. Rev. Nutr. 16:99-119 (1996).
P. Gunning, P. Ponte, H. Okayama, J. Engel, H. Blau, and L. Kedes. Isolation and characterization of full-length cDNA clones for human alpha-, beta-, and gamma-actin mRNAs: Skeletal but not cytoplasmic actins have an amino-terminal cysteine that is subsequently removed. Mol. Cell. Biol. 3:787-795 (1983).
A. Pavlova, E. Boutin, G. Cunha, and D. Sassoon. Msx1 (Hox-7.1) in the adult mouse uterus: Cellular interactions underlying regulation of expression. Development 120:335-345 (1994).
Y. Miyamoto, J. L. Coone, V. Ganapathy, and F. H. Leibach. Distribution and properties of the glycylsarcosine-transport system in rabbit renal proximal tubule: Studies with isolated brush-border-membrane vesicles. Biochem. J. 249:247-253 (1988).
S. Silbernagl, V. Ganapathy, and F. H. Leibach. H+gradient-driven dipeptide reabsorption in proximal tubule of rat kidney: Studies in vivoand in vitro. Am. J. Physiol. 253:F448-F457 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smith, D.E., Pavlova, A., Berger, U.V. et al. Tubular Localization and Tissue Distribution of Peptide Transporters in Rat Kidney. Pharm Res 15, 1244–1249 (1998). https://doi.org/10.1023/A:1011996009332
Issue Date:
DOI: https://doi.org/10.1023/A:1011996009332