Abstract
Purpose. The feasibility of using hydrophobicity measurements as screens for intracellular availability in T-cells or intestinal permeability in Caco-2 cells was examined.
Methods. T-cell experiments: Cells were counted, collected, then incubated with drug solution at 37°C. At selected time intervals, uptake was quenched by transferring a sample into oil, followed by rinsing, lysis of cells, protein precipitation and analysis by HPLC. Caco-2 cell experiments: Cells were grown on plastic dishes for 7−10 d, then rinsed and incubated with drug solution at 37°C. Uptake was quenched, cells were lysed, protein precipitated and drug was analyzed by HPLC. IAM chromatography: Stock solutions were injected onto an IAM column for HPLC. Mobile phase consisted of varying amounts of acetonitrile in buffer (pH 7.4). The capacity factor, k′IAM, was calculated using citric acid to measure the void volume and was obtained by extrapolation to pure buffer.
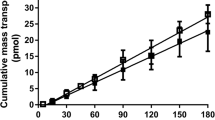
Results. Nine HIV protease inhibitors were studied for uptake by CEM T-cell suspensions or Caco-2 cell monolayers. Capacity factors (log) between IAM and C-18 columns were positively correlated for this series. Caco-2 uptake rates correlated well with T-cell uptake rates when normalized by protein mass. Single-variable regression using IAM or C-18 columns was acceptable for analysis of T-cell data. Correlation coefficients between T-cell uptake and log k′IAM or log k′C-18 were not improved with multivariable regression. Correlation between Caco-2 uptake and log k′IAM was enhanced when molecular weight and hydrogen-bonding potential were included in multivariable regression analysis (from r2 of 0.39 to 0.91). Correlations obtained using log k′IAM, log k′C-18.or log distribution coefficient (log D) were comparable when regressed against Caco-2 uptake using this approach. Calculated log partition coefficient (ClogP) provided the poorest correlation in the multivariable analysis (r2 = 0.57 for T-cell uptake and r2 = 0.71 for Caco-2 cell uptake).
Conclusions. Uptake of HIV protease inhibitors by T-cell suspensions or Caco-2 cell monolayers was positively correlated. Uptake by T-cell suspensions was adequately described by hydrophobicity alone. Description of uptake by Caco-2 cell monolayers required multivariable regression analysis in which molecular weight and hydrogen bonding were included. Experimental measures of hydrophobicity (log k′IAM, log k′C18 and log D) were superior to ClogP in the correlation analysis.
Similar content being viewed by others
REFERENCES
D. A. Paterson, R. A. Conradi, A. R. Hilgers, T. J. Vidmar, and P. S. Burton. A Non-aqueous partitioning system for predicting the oral absorption potential of peptides. Quant. Struct.-Act. Relat. 13:4–10 (1994).
K. B. Sentell and J. G. Dorsey. Retention mechanisms in reversed-phase liquid chromatography. Stationary-phase bonding density and partitioning. Anal. Chem. 61:930–934 (1989).
G. V. Betageri, A. Nayernama, and M. J. Habib. Thermodynamics of partitioning of nonsteroidal anti-inflammatory drugs in the N-octanol/buffer and liposome systems. Int. J. Pharm. Adv. 1:310–319 (1996).
C. Y. Yang, S. J. Cai, H. Liu, and C. Pidgeon. Immobilized artificial membranes-screens for drug membrane interactions. Adv. Drug Del. Rev. 23:229–56 (1996).
R. Kaliszan, A. Kaliszan, and I. W. Wainer. Deactivated hydrocarbonaceous silicia and immobilized artificial membrane stationary phases in high-performance liquid chromatographic determination of hydrophobicities of organic bases: relationship to log P and CLOGP. J. Pharm. Biomed. Anal. 11:505–11 (1993).
V. Pliska, B. Testa, and H. van de Waterbeemd. Lipophilicity in Drug Action and Toxicology. VCH, New York, 1996.
C. Pidgeon and U. V. Venkataram. Immobilized artificial membranes: chromatography supports composed of membrane lipids. Anal. Biochem. 176:36–47 (1989).
N. A. Roberts, J. A. Martin, D. Kinchington, A. V. Broadhurst, J. C. Craig, I. B. Duncan, S. A. Galpin, B. K. Handa, J. Kay, A. Krohn, R. W. Lambert, J. H. Merrett, J. S. Mills, K. E. B. Parkes, S. Redshaw, A. J. Ritchie, D. L. Taylor, G. J. Thomas, and P. J. Machin. Rational design of peptide-based HIV proteinase inhibitors. Science 248:358–61 (1990).
P. D. Brown and F. V. Sepulveda. A Rabbit jejunal isolated enterocyte preparation suitable for transport studies. J. Physiol. 363:257–270 (1985).
W. D. Stein. Theoretical and Experimental Biology An International Series of Monographs: The Movement of Molecules Across Cell Membranes, Academic Press, New York, 1967.
A. J. Leo. Hydrophobic parameter: measurement and calculation. In J. J. Langone (ed.), Methods in Enzymology, vol. 202A, Academic Press, New York, 1991, pp. 544–91.
F. H. Clarke and N. M. Cahoon. Potentiometric determination of the partition and distribution coefficients of dianionic compounds. J. Pharm. Sci. 84:53–54 (1995).
H. Lennernas, S. Nylander, and A.-L. Ungell. Jejunal permeability: a comparison between the Ussing chamber technique and the single-pass perfusion in humans. Pharm. Res. 14:667–671 (1997).
P. S. Burton, R. A. Conradi, N. F. H. Ho, A. R. Hilgers, and R. T. Borchardt. How structural features influence the biomembrane permeability of peptides. J. Pharm. Sci. 85:1336–40 (1996).
R. A. Conradi, A. R. Hilgers, N. F. H. Ho, and P. S. Burton. The influence of peptide structure on transport across Caco-2 cells. Pharm. Res. 8:1453–60 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stewart, B.H., Chung, F.Y., Tait, B. et al. Hydrophobicity of HIV Protease Inhibitors by Immobilized Artificial Membrane Chromatography: Application and Significance to Drug Transport. Pharm Res 15, 1401–1406 (1998). https://doi.org/10.1023/A:1011901605214
Issue Date:
DOI: https://doi.org/10.1023/A:1011901605214