Abstract
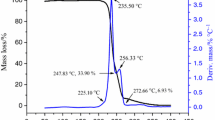
The thermal behavior of[Eu2(BA)6(dmbpy)2] (BA=C7H5O −2 , benzoate; dmbpy=C12H12N2, 4,4′-dimethyl-2,2′-bipyridine) and its kinetics were studied under the non-isothermal condition in a static air atmosphere by TG-DTG, IR and SEM methods. Thermal decomposition of [Eu2(BA)6(dmbpy)2] occurred in four consecutive stages at T P 232, 360, 455 and 495°C. The kinetic parameters were obtained from analysis of the TG-DTG curves by Achar and Madhusudanan—Krishnan—Ninan (MKN) methods. The most probable mechanisms for the first stage was suggested by comparing the kinetics parameters.
Similar content being viewed by others
References
Y. Zhang, L. P. Jin and S. Z. Lü, J. Inorg. Chem., (in Chinese), 13 (1997) 280.
L. P. Jin, R. F. Wang and L. S. Li, Polyhedron, 18 (1998) 487.
J. J. Zhang, R. F. Wang and J. B. Li, J. Therm. Anal. Cal., 62 (2000) 747.
J. J. Zhang, R. F. Wang and X. L. Zhai, Chinese J. Inorg. Chem., 16 (2000) 103.
R. F. Wang, L. P. Jin and L. S. Li, J. Coord. Chem., 47 (1999) 279.
B. N. Achar, Proc. Int. Clay Conf. Jerusalem, 1 (1964) 67.
P. M. Madhusudhanan, K. Krishnan and K. N. Ninan, Thermochim. Acta, 7 (1986) 189.
J. J. Zhang, L. G. Ge and X. L. Zhang, J. Therm. Anal., Cal., 58 (1999) 269.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J.J., Wang, R.F., Li, J.B. et al. Thermal Decomposition of (4,4′-Dimethyl-2,2′-Bipyridine) Tris(Benzoate) Europium(III): Non-isothermal kinetics. Journal of Thermal Analysis and Calorimetry 65, 241–248 (2001). https://doi.org/10.1023/A:1011553324335
Issue Date:
DOI: https://doi.org/10.1023/A:1011553324335