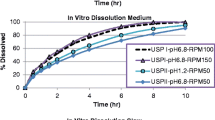
Abstract
The purpose of in vivo–in vitro correlation (IVIVC) modeling is described. These models are usually fitted to deconvoluted data rather than the raw plasma drug concentration/time data. Such a two-stage analysis is undesirable because the deconvolution step is unstable and because the fitted model predicts the fraction of a dosage unit dissolved/absorbed in vivo which generally is not the primary focus of our attention. Interest usually centers on the plasma drug concentration or some function of it (e.g., AUC, C max ). Incorporation of a convolution step into the model overcomes these difficulties. Odds, hazards, and reversed hazards models which include a convolution step are described. The identity model (which states that average in vivo and in vitro dissolution/time curves are coincident or directly superimposable) is a special case of these models. The odds model and the identity model were fitted to data sets for two different products using nonlinear mixed effects model fitting software. Results show that the odds model describes both data sets reasonably well and is a significantly better fit than the identity model in each case.
Similar content being viewed by others
REFERENCES
Food and Drug Administration. Guidance for Industry, Extended Release Oral Dosage Forms; Development, Evaluation and Application of In Vitro/In Vivo Correlations. Food and Drug Administration, Rockville, Md., 1997.
European Agency for the Evaluation of Medicinal Products. Note for Guidance on Quality of Modified Release Products: A: Oral Dosage Forms B: Transdermal Dosage Forms Section I (Quality), European Agency for the Evaluation of Medicinal Products, London, U.K., 1999.
J. Devane. Impact of IVIVR on product development. In D. Young, J. Devane, and J. Butler (eds.), Advances in Experimental Medicine and Biology, Vol. 423, In Vitro–In Vivo Correlations, Plenum Press, New York, 1997, pp. 241–259.
D. A. Piscitelli and D. Young. Setting dissolution specifications for modified-release dosage forms. In D. Young, J. Devane, and J. Butler (eds.), Advances in Experimental Medicine and Biology, Vol. 423, In Vitro–In Vivo Correlations, Plenum Press, New York, 1997, pp. 159–166.
United States Pharmacopeia. In Vitro and In Vivo Evaluation of Dosage Forms. Pharmacop. Forum 19:5366–5379 (1993).
Z. Hussein and M. Friedman. Release and absorption characteristics of novel theophylline sustained-release formulations. In vitro–in vivo correlation. Pharm. Res. 7:1167–1171 (1990).
P. Mojaverian, E. Radwanski, C. C. Lin, P. Cho, W. A. Vadino, and J. M. Rosen. Correlation of in vitro release rate and in vivo absorption characteristics of four chlorpheniramine maleate extended-release formulations. Pharm. Res. 9:450–456 (1992).
J. M. Cardot and E. Beyssac. In vitro/ in vivo correlations: Scientific implications and standardization. Eur. J. Drug Metab. Pharmacokin. 8:113–120 (1983).
S. S. Hwang, J. Gorsline, J. Louie, D. Dye, D. Guinta, and L. Hamel. In vitro and in vivo evaluation of a once-daily controlled-release pseudoephedrine product. J. Clin. Pharmacol. 35:259–267 (1995).
A. Dunne, T. O'Hara, and J. Devane. Level A in vivo– in vitro correlation: Nonlinear models and statistical methodology. J. Pharm. Sci. 86:1245–1249 (1997).
A. Dunne, T. O'Hara, and J. Devane, J. A new approach to modeling the relationship between in vitro and in vivo drug dissolution absorption. Statist. Med. 18:1865–1876 (1999).
J. Gabrielsson and D. Weiner. Pharmacokinetic and Pharmacodynamic Data Analysis: Concepts and Applications, Swedish Pharmaceutical Press, Sweden, 1997.
G. M. Wing. A Primer on Integral Equations of the First Kind—The Problem of Deconvolution and Unfolding, Society for Industrial and Applied Mathematics, Philadelphia, 1991.
W. R. Gillespie. Convolution-based approaches for in vivo–in vitro correlation modeling. In Advances in Experimental Medicine and Biology, Vol. 423, In Vitro–In Vivo Correlations, Plenum Press, New York, 1997, pp. 53–65.
A. Agresti. Categorical Data Analysis, Wiley, New York, 1990.
E. T. Lee. Statistical Methods for Survival Data Analysis, Wiley, New York, 1992.
M. Shaked and J. G. Shantikumar. Stochastic Orders and their Applications, Academic Press, London, 1994.
S. L. Beal and L. B. Sheiner. NONMEM User's Guides, NONMEM Project Group, University of California, San Francisco, 1992.
M. Pitsiu, G. Sathyan, S. Gupta, and D. Verotta. A semi-parametric deconvolution model to establish in in vivo–in vitro correlation applied to OROS Oxybutynin. J. Pharm. Sci. (in press).
C. Caramella, F. Ferrari, M. C. Bonferron, M. E. Sangalli, M. De Bernardi Di Valserra, F. Feletti, and M. R. Galmozzi. In vitro–in vivo correlation of prolonged release dosage forms containing diltiazem HCl. Biopharm. Drug. Dispos. 14:143–160 (1993).
H. Humbert, M. D., Cabiac, and H. Bosshardt. In vitro–in vivo correlation of a modified-release oral form of ketotifen: In vitro dissolution rate specification. J. Pharm. Sci. 83:131–136 (1994).
J. E. Polli, J. R. Crison, and G. L. Amidon. Novel approach to the analysis of In vitro–in vivo relationships. J. Pharm. Sci. 85:753–759 (1996).
Rights and permissions
About this article
Cite this article
O'Hara, T., Hayes, S., Davis, J. et al. In Vivo–In Vitro Correlation (IVIVC) Modeling Incorporating a Convolution Step. J Pharmacokinet Pharmacodyn 28, 277–298 (2001). https://doi.org/10.1023/A:1011531226478
Issue Date:
DOI: https://doi.org/10.1023/A:1011531226478