Abstract
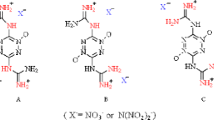
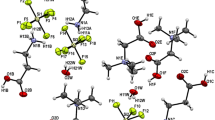
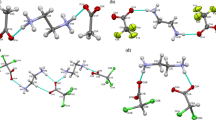
The crystal structures of two amine base salts, the hydrazinium, 1, and the hydroxylammonium, 2, of dinitramide have been determined. 1 crystallizes in the monoclinic space group P 21/c with cell dimensions a = 8.312(3), b = 5.654(1), c = 10.659(3) Å, β = 93.73(3)°, while 2 crystallizes in the orthorhombic space group Pcab (nonstandard setting of Pbca) with cell dimensions a = 6.439(2), b = 12.470(4), c = 30.816(14) Å. The structures of 1 and 2 contain protonated amine cations and dinitramide anions linked by hydrogen bonding. In addition, in 2 there are both neutral and zwitterionic hydroxylamine moieties involved in the hydrogen bonding scheme. Thus in 2 the complete formula unit is (NH3 +OH)2[N3O4 −]2 · (NH2OH) middot; (NH3 +O−), and in this structure the hydroxylamine exists in its three possible forms: protonated, neutral, and zwitterionic. In both structures the conformations adopted by the dinitramine anions can be related to the types of hydrogen bonds it forms with the surrounding amine cations.
Similar content being viewed by others
References
Sitzman, M.E.; Gilardi, R.; Butcher, R.J.; Koppes, W.M.; Stern A.G.; Trasher, J.S.; Trivedi, N. J.; Yang, Z-Y. Inorg. Chem. 2000, 39, 843.
Butcher, R.J.; Gilardi, R. Thermochim. Acta 2001, in press.
Politzer, P.; Seminario, J.M. Chem. Phys. Lett. 1993, 216, 348.
Michels, H.H.; Montgomery Jr., J.A. J. Phys. Chem. 1993, 97, 6602.
Mebel, A.M.; Lin, M.C.; Morokuma, K.; Melius, C.F. J. Phys. Chem. 1995, 99, 6842.
Leroy, G.; Sana, M.; Wilante, C.; Peeters, D.; Dogimont, C. J. Mol. Struct. (THEOCHEM), 1987, 153, 249.
Leroy, G.; Sana, M.; Wilante, C.; Peeters, D.; Bourasseau, S. J. Mol. Struct. (THEOCHEM), 1989, 187, 251.
Brinck, T.; Murray, J.S.; Politzer, P. J. Org. Chem. 1991, 56, 5012.
Martin, A.; Pinkerton, A.A.; Gilardi, R.D.; Bottaro, J.C. Acta Crystallogr. 1997, B53, 504.
Trammell, S.; Goodson, P.A.; Sullivan, B.P. Inorg. Chem. 1996, 35, 1421.
Gilardi, R.; Flippen-Anderson, J.; George, C.; Butcher, R.J. J. Am. Chem. Soc. 1997, 119, 9411, and references therein.
Butcher, R.J.; Gilardi, R. J. Chem. Crystallogr. 1998, 28, 95.
Butcher, R.J.; Gilardi, R. J. Chem. Crystallogr. 1998, 28, 163.
Butcher, R.J.; Gilardi, R. J. Chem. Crystallogr. 1998, 28, 105.
Butcher, R.J.; Gilardi, R. J. Chem. Crystallogr. 1998, 28, 673.
Tanbug, R.; Kirschbaum, K.; Pinkerton, A.A. J. Chem. Crystallogr. 1999, 29, 45, and references therein.
Hardie, M.J.; Martin, A.; Pinkerton, A.A.; Zhurova, E. A. Acta Crystallogr. 2001, B57, 113.
Dubovitskii, F.I.; Golovina, N.I.; Pavlov, A.N.; Atovmyan, L.O. Dokl. Akad. Nauk. SSSR, 1998, 360, 491.
Sheldrick, G.M. SHELXTL,Crystallographic System; Siemens Analytical Instrument Division, Madison, WIS, 1996.
Cromer, D.T.; Waber J.T. International Tables for X-Ray Crystallography, Vol IV; The Kynoch Press, Birmingham, England, 1974.
Cromer, D.T. International Tables for X-Ray Crystallography, Vol IV; The Kynoch Press, Birmingham, England, 1974.
Trueblood, K.N.; Knobler, C.B.; Lawrence, D.S.; Stevens, R.V. J. Am. Chem. Soc. 1982, 104, 1355.
Henschel, D.; Wijaya, K.; Jones, P.G.; Blaschette, A. Acta Crstallogr. Sect. C (Cr. Str. Comm.) 1999, 55, 664.
Schreuer, J.Z. Kristallogr.-New Crystal Structures 1999, 214, 319.
Benniston, A.C.; Yufit, D.S.; Howard, J.K. Acta Crystallogr. Sect. C (Cr. Str. Comm.) 1999, 55, 1535.
Gajapathy, D.; Govindarajan, S.; Patil, K.C.; Manohar, H. Polyhedron 1983, 2, 865.
Chekhlov, A.N.; Martynov, I.V.; Brel, V.K. Dokl. Akad. Nauk. SSSR 1987, 292, 149.
Kickham, J.E.; Loeb, S. J. Chem. Commun. 1993, 1848.
Ahmed, N.A.K.; Liminga, R.; Olovsson, I. Acta Chem. Scand. 1968, 22, 88.
Thomas, J.O. Acta Crystallogr. Sect B 1973, 29, 1767.
Thomas, J.O.; Liminga, R. Acta Crystallogr. Sect B 1978, 34, 686.
Dickens, B. J. Res. Nat. Bur. Stand. A 1970, 74, 309.
Hady, S.A.; Nahringbauer, I.; Olovsson, I. Acta Chem. Scand. 1969, 23, 2764.
Garcia, E.; Lee, K-Y.; Storm, C.B. Acta Crystallogr. Sect. C (Cr. Str. Comm.) 1992, 48, 1682.
Bircher, H.-R.; Ochsenbein, P.; Hauser, J.; Burgi, H.-B. Acta Crystallogr. Sect. C (Cr. Str. Comm.) 1996, 52, 2002.
Gusev, A.I.; Nesterov, D. Yu.; Zhigach, A.F.; Svitzin, R.A.; Sobolev, E.S. Zh. Strukt. Khim. 1978, 19, 180.
Sellman, D.; Freidrich, H.; Knoch, F.Z. Naturforsch. Teil B 1994, 49, 660.
Fun, H-K.; Sivakumar, K.; Jiang, Y-Z.; Sun, J.; Zhou, Z-Y. Acta Crystallogr. Sect. C (Cr. Str. Comm.) 1995, 51, 2085.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gilardi, R., Butcher, R.J. A new class of flexible energetic salts, part 6: the structures of the hydrazinium and hydroxylammonium salts of dinitramide. Journal of Chemical Crystallography 30, 599–604 (2000). https://doi.org/10.1023/A:1011362427996
Issue Date:
DOI: https://doi.org/10.1023/A:1011362427996