Abstract
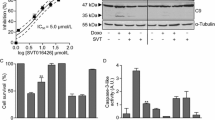
Phosphatidylserine (PS) is exposed on the outer leaflet of the plasma membrane in apoptotic cell death. However, the roles of PS in apoptotic signaling are still unclear. In this study, we found that exogenous PS, but not other phospholipids, induced cell death in adherent cells, but not in suspension culture. The cell death exhibited typical features of apoptosis such as cell shrinkage, nuclear fragmentation and abnormal chromatin condensation. When PS was added to CHO-K1 cells in monolayer culture, they began to show changes in cell shape and actin cytoskeleton and protein kinase C (PKC) activity, followed by cell detachment, caspase activation, cleavage of focal adhesion kinase (FAK) and finally loss of viability. These results suggested that PS causes apoptosis through actin disorganization, cell detachment and cleavage of FAK.
Similar content being viewed by others
References
Raff MC. Social controls on cell survival and cell death. Nature 1992; 356: 397-400.
Wyllie AH, Kerr JFR, Currie AR. Cell death: The significance of apoptosis. Int Rev Cytol 1980; 68: 251-306.
Fadok VA, Voelker DR, Campbell PA, Cohen JJ, Bratton DL, Henson PM. Exposure of phosphatidylserine on the cell surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol 1992; 148: 2207-2216.
Zachowski A. Phospholipids in animal eukaryotic membranes: transverse asymmetry and movement. Biochem J 1993; 294: 1-14.
Op den Kamp JAF. Lipid asymmetry in membranes. Annu Rev Biochem 1979; 48: 47-71.
Bratton DL, Fadok VA, Richter DA, Kailey JM, Guthrie LA, Henson PM. Appearance of phosphatidylserine on apoptotic cells requires calcium-mediated nonspecific flip-flop and is enhanced by loss of the aminophospholipid translocase. J Biol Chem 1997; 272: 26159-26165.
Zhao J, Zhou Q, Wiedmer T, Sims PJ. Level of expression of phospholipid scramblase regulates induced movement of phosphatidylserine to the cell surface. J Biol Chem 1998; 273: 6603-6606.
Shiratsuchi A, Osada S, Kanazawa S, NakanishiY. Essential role of phosphatidylserine externalization in apoptosing cell phagocytosis by macrophages. Biochem Biophys Res Commun 1998; 246: 549-555.
Nishizuka Y. The role of protein kinase C in cell surface signal transduction and tumor promotion. Nature 1984; 308: 693-698.
Ghosh S, Xie WQ, Quest AFG, Mabrouk GM, Strum JC, Bell RM. The cysteine-rich region of raf-1 kinase contains zinc, translocates to liposomes, and is adjacent to a segment that binds GTP-ras. J Biol Chem 1994; 269: 10000-10007.
Calderon C, Huang Z-H, Gage DA, Sotomayor EM, Lopez DM. Isolation of a nitric oxide inhibitor from mammary tumor cells and its characterization as phosphatidyl serine. J Exp Med 1994; 180: 945-958.
Hebdon GM, LeVine H, Sahyoun NE, Schmitges CJ, Cuatrecasas P. Specific phospholipids are required to reconstitute adenylate cyclase solubilized from rat brain. Proc Natl Acad Sci USA 1981; 78: 120-123.
Kuge O, Nishijima M, Akamatsu Y. Phosphatidylserine biosynthesis in cultured Chinese hamster ovary cells. III. Genetic evidence for utilization of phosphatidylcholine and phosphatidylethanolamine as precursors. J Biol Chem 1986; 261: 5795-5798.
Aussel C, Pelassy C, Breittmayer JP. CD95 (Fas/APO-1) induces an increased phosphatidylserine synthesis that precedes its externalization during programmed cell death. FEBS Lett 1998; 431: 195-199.
Uchida K, Emoto K, Daleke DL, Inoue K, Umeda M. Induction of apoptosis by phosphatidylserine.J Biochem 1998; 123: 1073-1078.
Iguchi K, Usami Y, Hirano K, Hamatake M, Shibata M, Ishida R. Decreased thymosin ?4 in apoptosis induced by a variety of antitumor drugs. Biochem Pharmacol 1999; 57: 1105-1111.
Stefanis L, Park DS, Yan CYI, et al. Induction of CPP-32-like activity in PC12 cells by withdrawal of tropic support. J Biol Chem 1996; 271: 30633-30671.
Bradford MM. A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976; 72: 248-254.
Yasuda I, Kishimoto A, Tanaka S, Tominaga M, Sakurai A, Nishizuka Y. A synthetic peptide substrate for selective assay of protein kinase C. Biochem Biophys Res Commun 1990; 166: 1220-1227.
Wen LP, Fahrni JA, Troie S, Guan JL, Orth K, Rosen GD. Cleavage of focal adhesion kinase by caspases during apoptsis. J Biol Chem 1997; 272: 26056-26061.
Endo TA, Kobayashi T, Ohki K. A Chinese hamster ovary cell mutant resistant to phosphatidylserine is defective in transbilayer movement of cell surface phosphatidylserine. Exp Cell Res 1996; 228: 341-346.
Frisch SM, Francis H. Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol 1994; 124: 619-626.
Meredith JE Jr, Fazeli B, Schwart MA. The extracellular matrix as a cell survival factor. Mol Biol Cell 1993; 4: 953-961.
Water Bob van de, Nagelkerke JF, Stevens JL. Dephosphorylation of focal adhesion kinase (FAK) and loss of focal contacts precede caspase-mediated cleavage of FAK during apoptosis in renal epithelial cells. J Biol Chem 1999; 274: 13328-13337.
Downward J. Mechanisms and consequences of activation of protein kinase B/Akt. Curr Opin Cell Biol 1998; 10: 262-267.
Fujio Y, Walsh K. Akt mediates cytoprotection of endothelial cells by vascular endothelial growth factor in an anchorage-dependent manner. J Biol Chem 1999; 274: 16349-16354.
Kornberg L, Earp HS, Parsons JT, Schaller M, Juliano RL. Cell adhesion or integrin clustering increases phosphorylation of a focal adhesion-associated tyrosine kinase. J Biol Chem 1992; 267,23439-23442.
Schaller MD, Borgman CA, Cobb BS, Vines RR, Reynolds AB, Parsons JT. pp125FAK a structurally distinctive protein-tyrosine kinase associated with focal adhesions. Proc Natl Acad Sci USA 1992; 89: 5192-5196.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Iguchi, K., Hirano, K., Hamatake, M. et al. Phosphatidylserine induces apoptosis in adherent cells. Apoptosis 6, 263–268 (2001). https://doi.org/10.1023/A:1011331424311
Issue Date:
DOI: https://doi.org/10.1023/A:1011331424311