Abstract
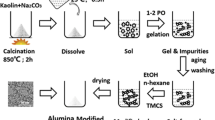
The effect of wetting non-hydrolytic derived alumina xerogels with water and organic solvents in the 20–70°C range on the alumina's properties was investigated. Wetting with organic solvents does not affect the alumina. However, contact with water was found to change the sharp crystallization at ∼800°C to a continuous crystallization starting at ∼450°C. Water treatment for a day at room temperature (RT) followed by second calcination decreased the surface area by 10%. This decrease in surface area is less pronounced with increasing wetting periods. On the other hand water treatment at 50–70°C followed by a second calcination resulted in a surface area increase of up to 15%. Upon water treatment the total pore volume has decreased from 0.65 (cm3/gr) to 0.48 (cm3/gr) and the average pore size decreased from 6.8 nm to 4.1 nm. The Cl content was found to be uneffected by the water treatment, remaining at ∼2.5% wt. Wetting with water at elevated temperature (70°C) accelerated the morphological changes, eliminating the crystallization peak at 800°C in one hour. A dissolution-reprecipitation mechanism is suggested to explain the results. In addition, Mass-Spectroscopy of the effluent gas during heat treatment revealed the emission of CO2 and water upon phase transition into α-Al2O3, at 1150–1300°C.
Similar content being viewed by others
References
R.K. Oberlander, in Applied Industrial Catalysis V(3), edited by B.E. Leach (Academic Press, London, 1984), p. 63.
B.E. Yoldas, J. Appl. Chem. Biotechnol. 23, 803 (1973).
C.J. Brinker and G.W. Scherer, Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing (Academic Press, New York, 1990).
Y. de Hazan, G.E. Shter, Y. Cohen, C. Rottman, D. Avnir, and G.S. Grader, J. Sol-Gel Sci. Technol. 14(3), 233 (1999).
G.S. Grader, Y. de Hazan, D.B. Zhivotovskii, and G.E. Shter, J. Sol-Gel Sci. Techl. 10, 127 (1997).
S. Acosta, R. Corriu, D. Leclercq, P.H. Mutin, and A. Vioux, J. Sol-Gel Sci. Tech. 2, 25 (1994).
S. Acosta, P. Arnal, R.J.P. Corriu, D. Leclercq, P.H. Mutin, and A. Vioux, in Better Ceramics through Chemistry VI, Mater. Res. Soc. Symp. Proc. 346, 43 (1994).
S. Acosta, R.J.P. Corriu, D. Leclercq, P. Lefevre, P.H. Mutin, and A. Vioux, J. Non-Cryst. Solids 170, 234 (1994).
M. Andrianainarivelo, R.J.P. Corriu, D. Leclercq, P.H. Mutin, and A. Vioux, J. Mater. Chem. 7(2), 279 (1997).
M. Andrianainarivelo, R.J.P. Corriu, D. Leclercq, P.H. Mutin, and A. Vioux, Chem. Mater. 9(5), 1098 (1997).
M. Andrianainarivelo, R. Corriu, D. Leclercq, P.H. Mutin, and A. Vioux, J. Mater. Chem. 6(10), 1665 (1996).
J.T. Richardson, Principles of Catalyst Development (Plenum Press, New York, 1989).
S.Y. Lee and R. Aris, Catal. Rev. 27(2), 207 (1985).
J.H. de Boer, Angew. Chem. 64, 563 (1952).
L. Jacimovic, J. Stevovic, and S. Veljkovic, J. Phys. Chem. 76(24), 3625 (1972)
R.W. Maatman. P. Mahaffy, P. Hoekstra, and C. Addink, J. Catal. 23, 105 (1971).
H. Knozinger and P. Ratnasamy, Catal. Rev. 17(1), 31 (1978).
J.B. Peri, J. Phys. Chem. 69(1), 220 (1965).
D. Maret, G.M. Pajonk, and S.J. Teichner, in Spillover of Adsorbed Species, edited by G.M. Pajonk, S.J. Teichner, and J.E. Germain (Elsevier Science, Amsterdam, 1983), p. 215.
G.C. Bye and J.G. Robinson, Koll. Z., and Z. Polym. 198(1/2), 53 (1964).
J.A. Dyer, N.C. Scrivner, and S.K. Dental, Environ. Prog. 17(1), 1 (1998).
Y. de Hazan, Synthesis and characterization of alumina gel, Ph.D. Dissertation, Technion, Haifa, 1998.
J.P. Brunelle, Pure & Appl. Chem. 50, 1211 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grader, G., Shter, G., Avnir, D. et al. Modification of Non-Hydrolytic Sol-Gel Derived Alumina by Solvent Treatments. Journal of Sol-Gel Science and Technology 21, 157–165 (2001). https://doi.org/10.1023/A:1011266100079
Issue Date:
DOI: https://doi.org/10.1023/A:1011266100079