Abstract
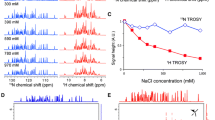
TROSY-type optimization of liquid-state NMR experiments is based on the preservation of unique coherence transfer pathways with distinct transverse relaxation properties. The broadband decoupling of the 1H spins interchanges the TROSY and anti-TROSY magnetization transfer pathways and thus is not used in TROSY-type triple resonance experiments or is replaced with narrowband selective decoupling. To achieve the full advantage of TROSY, the uniform deuteration of proteins is usually required. Here we propose a new and general method for 1H broadband decoupling in TROSY NMR, which does not compromise the relaxation optimization in the 15N–1H moieties, but uniformly and efficiently refocuses the 1 J CH scalar coupling evolution in the 13C–1H moieties. Combined with the conventional 2H decoupling, this method enables obtaining high sensitivity TROSY-type triple resonance spectra with partially deuterated or fully protonated 13C,15N labeled proteins.
Similar content being viewed by others
References
Canet, D. (1989) Prog. NMR Spectrosc., 21, 237-291.
Cavanagh, J. and Rance, M. (1992) J. Magn. Reson., 96, 670-678.
Chook, Y.M., Gray, J.V., Ke, H.M. and Lipscomb, W.N. (1994) J. Mol. Biol., 240, 476-500.
Gardner, K.H. and Kay, L.E. (1998) Annu. Rev. Biophys. Biomol. Struct., 27, 357-406.
Goldman, M. (1984) J. Magn. Reson., 60, 437-452.
Grzesiek, S. and Bax, A. (1993) J. Am. Chem. Soc., 115, 12593-12594.
Grzesiek, S., Wingfield, P., Stahl, S., Kaufman, J.D. and Bax, A. (1995) J. Am. Chem. Soc., 117, 9594-9595.
Gschwind, R.M., Kessler, H. and Gemmecker, G. (1999) J. Magn. Reson., 137, 285-288.
Kay, L.E., Keifer, P. and Saarinen, T. (1992) J. Am. Chem. Soc., 114, 10663-10665.
Ladner, J.E., Reddy, P., Davis, A., Tordova, M., Howard, A.J. and Gilliland, G.L. (2000) Acta Crystallogr., D56, 673-683.
LeMaster, D.M. (1994) Prog. NMR Spectrosc., 26, 371-419.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989) J. Magn. Reson., 85, 393-399.
Matsuo, H., Kupce, E., Li, H.J. and Wagner, G. (1996) J. Magn. Reson., 113, 91-96.
Pervushin, K., Riek, R., Wider, G. and Wüthrich, K. (1997) Proc. Natl. Acad. Sci. USA, 94, 12366-12371.
Pervushin, K., Wider, G. and Wüthrich, K. (1998) J. Biomol. NMR, 12, 345-348.
Salzmann, M., Pervushin, K., Wider, G., Senn, H. and Wüthrich, K. (1998) Proc. Natl. Acad. Sci. USA, 95, 13585-13590.
Salzmann, M., Wider, G., Pervushin, K., Senn, H. and Wüthrich, K. (1999) J. Am. Chem. Soc., 121, 844-848.
Shaka, A.J., Keeler, J., Frenkiel, T. and Freeman, R. (1983) J. Magn. Reson., 52, 335-338.
Shan, X., Gardner, K.H., Muhandiram, D.R., Rao, N.S., Arrowsmith, C.H. and Kay, L.E. (1996) J. Am. Chem. Soc., 118, 6570-6579.
Shimizy, H. (1964) J. Phys. Chem., 40, 3357-3364.
Sobol, A.G., Wider, G., Iwai, H. and Wüthrich, K. (1998) J. Magn. Reson., 130, 262-271.
Stonehouse, J., Clowes, R.T., Shaw, G.L., Keeler, J. and Laue, E.D. (1995) J. Biomol. NMR, 5, 226-232.
Van der Kooi, C.W., Kupce, E., Zuiderweg, E.R.P. and Pellecchia, M. (1999) J. Biomol. NMR, 15, 335-338.
Yamazaki, T., Lee, W., Arrowsmith, C.H., Muhandiram, D.R. and Kay, L.E. (1994) J. Am. Chem. Soc., 116, 11655-11666.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eletsky, A., Kienhöfer, A. & Pervushin, K. TROSY NMR with partially deuterated proteins. J Biomol NMR 20, 177–180 (2001). https://doi.org/10.1023/A:1011265430149
Issue Date:
DOI: https://doi.org/10.1023/A:1011265430149