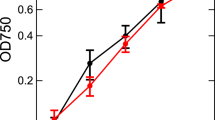
Abstract
We have used circular dichroism (CD) spectroscopy and chlorophyll fluorescence induction measurements in order to examine low-pH-induced changes in the chiral macro-organization of the chromophores and in the efficiency of non-photochemical quenching of the chlorophyll a fluorescence (NPQ) in intact, dark-adapted cells of Chlorella fusca (Chlorophyceae) and Mantoniella squamata (Prasinophyceae). We found that: (i) high proton concentrations enhanced the formation of chiral macrodomains of the complexes, i.e. the formation of large aggregates with long-range chiral order of pigment dipoles; this was largely independent of the low-pH-induced accumulation of de-epoxidized xanthophylls; (ii) lowering the pH led to NPQ; however, efficient energy dissipation, in the absence of excess light, could only be achieved if a substantial part of violaxanthin was converted to zeaxanthin and antheraxanthin in Chlorella and Mantoniella, respectively; (iii) the low-pH-induced changes in the chiral macro-organization of pigments were fully reversed by titrating the cells to neutral pH; (iv) at neutral pH, the presence of antheraxanthin or zeaxanthin did not bring about a sizeable NPQ. Hence, low-pH-induced NPQ in dark adapted algal cells appears to be associated both with the presence of de-epoxidized xanthophylls and structural changes in the chiral macrodomains. It is proposed that the macrodomains, by providing a suitable structure for long-distance migration of the excitation energy, in the presence of quenchers associated with de-epoxidized xanthophylls, facilitate significantly the dissipation of unused excitation energy.
Similar content being viewed by others
References
Avital S and Malkin S (1998) Quenching of chlorophyll fluorescence by carotenoids in a micellar model system. In: Garab G (ed) Photosynthesis: Mechanisms and Effects, Vol I, pp 477–482. Kluwer Academic Publishers, Dordrecht, The Netherlands
Barber J (1986) Surface electric charges and protein phosphorylation. In: Staehelin LA and Arntzen CJ (eds) Encyclopedia of Plant Physiology, Vol 19, pp 653–664. Springer Verlag, Berlin
Barzda V, Mustárdy L and Garab G (1994) Size dependency of circular dichroism in macroaggregates of photosynthetic pigment– protein complexes. Biochemistry 33: 10837–10841
Barzda V, Garab G, Gulbinas V and Valkunas L (1995) Long distance migration of excitation energy and fluorescence quenching mechanism in chiral macroaggregates of LHC II. In: Mathis P (ed) Photosynthesis: From Light to Biosphere, Vol I, pp 319–322. Kluwer Academic Publishers, Dordrecht, The Netherlands
Barzda V, Istokovics A, Simidjiev I and Garab G (1996a) Structural flexibility of chiral macroaggregates of light-harvesting chlorophyll a/b pigment–protein complexes. Light-induced reversible structural changes associated with energy dissipation. Biochemistry 35: 8981–8985
Barzda V, Garab G, Gulbinas V and Valkunas L (1996b) Evidence for long-range excitation migration in macroaggregates of the chlorophyll a/b light-harvesting antenna complexes. Biochim Biophys Acta 1273: 231–236
Barzda V, Jennings RC, Zucchelli G and Garab G (1999) Kinetic analysis of the light-induced fluorescence quenching in light-harvesting chlorophyll a/b pigment–protein complex of Photosystem II. Photochem Photobiol 70(5): 751–759
Cseh Z, Papp E and Garab G (1998) Model calculations on the mechanism of the light-induced structural changes in lamellar aggregates of LHC II. In: Garab G (ed) Photosynthesis: Mechanisms and Effects, Vol I, pp 345–348. Kluwer Academic Publishers, Dordrecht, The Netherlands
Demmig-Adams B (1990) Carotenoids and photoprotection in plants: a role for the xanthophyll zeaxanthin. Biochim Biophys Acta 1020: 1–24
Demmig-Adams B, Adams III WW, Heber U, Neimanis S, Winter K, Krüger A, Czygan FC, Bilger W and Björkman O (1990) Inhibition of zeaxanthin formation and of rapid changes in radiationless energy dissipation by dithiothreitol in spinach leaves and chloroplasts. Plant Physiol 92: 293–301
Demmig-Adams B and Adams III WW (1996) The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci 1: 21–26
Finzi L, Bustamante C, Garab G and Juang C-B (1989) Direct observation of large chiral domains in chloroplast thylakoid membranes by differential polarization microscopy. Proc Natl Acad Sci USA 86: 8748–8752
Frank HA, Cua A, Chynwat V, Young AJ, Gosztola D and Wasielewski MR (1994) Photophysics of the carotenoids associated with the xanthophyll cycle in photosynthesis. Photosynth Res 41: 389–395
Garab G (1992) Macrodomain organization of complexes in the thylakoid membranes. In: Murata N (ed) Research in Photosynthesis, Vol I, pp 171–178. Kluwer Academic Publishers, Dordrecht, The Netherlands
Garab G (1996) Linear and circular dichroism. In: Amesz J and Hoff AJ (eds) Biophysical Techniques in Photosynthesis, pp 11–40. Kluwer Academic Publishers, Dordrecht, The Netherlands
Garab G and Mustárdy L (1999) Role of LHC II-containing macrodomains in the structure, function and dynamics of grana. Aust J Plant Physiol 26: 649–658
Garab G, Faludi-Daniel A, Sutherland JC and Hind G (1988a) Macro-organization of chlorophyll a/b light-harvesting complex in thylakoids and aggregates: Information from circular differential scattering. Biochemistry 27: 2425–2430
Garab G, Leegood RC, Walker DA, Sutherland JC and Hind G (1988b) Reversible changes in macro-organization of the lightharvesting chlorophyll a/b pigment–protein complex detected by circular dichroism. Biochemistry 27: 2430–2434
Garab G, Wells KS, Finzi L and Bustamante C (1988c) Helically organized macroaggregates of pigment–protein complexes in chloroplasts: Evidence from circular intensity differential scattering. Biochemistry 27: 5839–5843
Garab G, Kieleczawa J, Sutherland JC, Bustamante C and Hind G (1991) Organization of pigment–protein complexes into macrodomains in the thylakoid membranes of wild-type and chlorophyll b-less mutant of barley as revealed by circular dichroism. Photochem Photobiol 54: 273–281
Garab G, Istokovics A, Butiuc A, Simidjiev I and Dér A (1998) Light-induced ion movements in thylakoid membranes and isolated LHC II. In: Garab G (ed) Photosynthesis: Mechanisms and Effects, Vol I, pp 341–344. Kluwer Academic Publishers, Dordrecht, The Netherlands
Gilmore AM (1997) Mechanistic aspects of xanthophyll cycledependent photoprotection in higher plant chloroplasts and leaves. Physiol Plant 99: 197–209
Gilmore AM and Yamamoto HY (1992) Dark-induction of zeaxanthin-dependent nonphotochemical fluorescence quenching mediated by ATP. Proc Natl Acad Sci USA 89: 1899–1903
Gilmore AM and Yamamoto HY (1993) Linear models relating xanthophylls and lumen acidity to non-photochemical fluorescence quenching: Evidence that antheraxanthin explains zexanthin-independent quenching. Photosynth Res 35: 67–78
Gilmore AM, Hazlett T and Govindjee (1995) Xanthophyll cycledependent quenching of Photosystem II chlorophyll a fluorescence: Formation of a quenching complex with a short fluorescence lifetime. Proc Natl Acad Sci USA 92: 2273–2277
Goss R, Richter M and Wild A (1995) Role of ΔpH in the mechanism of zeaxanthin-dependent amplification of qE. J Photochem Photobiol B, Biol 27: 147–152
Goss R, Böhme K and Wilhelm C (1998) The xanthophyll cycle of Mantoniella squamata converts violaxanthin to antheraxanthin but not to zeaxanthin: Consequences for the mechanism of enhanced non-photochemical energy dissipation. Planta 205: 613–621
Goss R, Wilhelm C and Garab G (2000) Organization of the pigment molecules in the chlorophyll a/b/c containing alga Mantoniella squamata (Prasinophyceae) studied by means of absorbance, circular and linear dichroism spectroscopy. Biochim Biophys Acta 1457: 190–199
Gruszecki WI, Grudzinski W, Banaszek-Glos A, Matula M, Kernen P, Krupa Z and Sielewiesiuk J (1999a) Xanthophyll pigments in light-harvesting complex II in monomolecular layers: Localisation, energy transfer and orientation. Biochim Biophys Acta 1412: 173–183
Gruszecki WI, Grudzinski W, Matula M, Kernen P and Krupa Z (1999b) Light-induced excitation quenching and structural transition in light-harvesting complex II. Photosynth Res 59: 175–185
Gülen D, Van Grondelle G and Van Amerongen H (1997) Structural information on the light harvesting complex II of green plants that can be deciphered from polarised absorption characteristics. J Phys Chem B 101: 7256–7261
Gussakovsky EE, Barzda V, Shahak Y and Garab G (1997) Irreversible disassembly of chiral macrodomains in thylakoids due to photoinhibition. Photosynth Res 51: 119–126
Hager A (1967) Untersuchungen über die lichtinduzierten reversiblen Xanthophyllumwandlungen in Chlorella und Spinacia. Planta 74: 148–172
Hager A (1969) Lichtbedingte pH-Erniedrigung in einem Chloroplasten-Kompartiment als Ursache der enzymatischen Violaxanthin-zu Zeaxanthin-Umwandlung. Beziehungen zur Photophosphorylierung. Planta 89: 224–243
Hase E and Morimuro Y (1971) Synchronous and homocontinuous cultures of algae. In: Pietro S (ed) Methods in Enzymology 23, pp 81–106. Academic Press, New York
Horton P, Ruban AV, Rees D, Pascal AA, Noctor G and Young AJ (1991) Control of the light harvesting function of chloroplast membranes by aggregation of the LHC II chlorophyll–protein complex. FEBS Lett 292: 1–4
Horton P, Ruban AV and Walters RG (1996) Regulation of light harvesting in green plants. Annu Rev Plant Physiol Plant Mol Biol 47: 655–684
Horton P (1999) Hypothesis: Are grana necessary for regulation of light harvesting? Aust J Plant Physiol 26: 659–669
Istokovics A, Simidjiev I, Lajko F and Garab G (1997) Characterization of the light-induced reversible changes in the chiral macro-organization of the chromophores in chloroplast thylakoid membranes. Temperature dependence and effect of inhibitors. Photosynth Res 54: 45–53
Jennings RC, Garlaschi FM and Zucchelli G (1991) Light-induced fluorescence quenching in the light-harvesting chlorophyll a/b protein complex. Photosynth Res 34: 375–385
Karukstis KK and Sauer K (1985) The effects of cation-induced and pH-induced membrane stacking on chlorophyll fluorescence decay kinetics. Biochim Biophys Acta 806: 374–388
Keller D and Bustamante C (1986a) Theory of the interaction of light with large inhomogeneous molecular aggregates. I. Absorption. J Chem Phys 84: 2961–2971
Keller D and Bustamante C (1986b) Theory of the interaction of light with large inhomogeneous molecular aggregates. II. Psi-type circular dichroism. J Chem Phys 84: 2972–2979
Kitmitto A, Mustafa AO, Ford JW, Holzenburg A and Ford RC (1999) Does photoinhibition and/or phosphorylation of Photosystem II influence its in vivo oligomeric state? Biochim Biophys Acta 1413: 21–30
Krämer P, Wilhelm C, Wild A, Mörschel E and Rhiel E (1988) Ultrastructure and freeze-fracture studies of thylakoids of Mantoniella squamata (Prasinophyceae). Protoplasma 147: 170–177
Krieger A and Weis E (1993) The role of calcium in the pHdependent control of photosystem II. Photosynth Res 37: 117–130
Li X-P, Björkman O, Shih C, Rosenquist M, Jansson S and Niyogi KK (2000) A pigment–binding protein essential for the regulation of photosynthetic light harvesting. Nature 403: 391–395
Müller D (1962) Ñber jahres-und lunarperiodische Erscheinungen bei einigen Braunalgen. Bot Mar 4: 140–155
Naqvi KR, Melo TB, Raju BB, Jávorfi T, Simidjiev I and Garab G (1997) Quenching of chlorophyll a singlets and triplets by carotenoids in the light-harvesting complex of Photosystem II: Comparison of aggregates with trimers. Spectrochim Acta Part A 53: 2659–2667
Naqvi KR, Jávorfi T, Melo TB and Garab G (1999) More on the catalysis of internal conversion in chlorophyll a by an adjacent carotenoid in light-harvesting complex (Chl a/b LHC II) of higher plants: Time-resolved triplet-minus-singlet spectra of detergent-perturbed complexes. Spectrochim Acta Part A 55: 193–204
Osuka A, Kume T, Haggquist GW, Jávorfi T, Lima JC, Melo E and Naqvi KR (1999) Photophysical characteristics of two model antenna systems: A fucoxanthin-pyropheophorbide dyad and its peridinin analogue. Chem Phys Let 313: 499–504
Pfündel E and Bilger W (1994) Regulation and possible function of the violaxanthin cycle. Photosynth Res 42: 89–109
Phillip D, Ruban AV, Horton P, Asato A and Young AJ (1996) Quenching of chlorophyll fluorescence in the major light harvesting complex of Photosystem II. Proc Natl Acad Sci USA 93: 1492–1497
Polivka T, Herek JL, Zigmantas D, Akerlund H-E and Sundström V (1999) Direct observation of the (forbidden) S1 state in carotenoids. Proc Natl Acad Sci USA 96:4914–4917
Rees D, Noctor G, Ruban AV, Crofts J, Young AJ and Horton P (1992) pH dependent chlorophyll fluorescence quenching in spinach thylakoids from light treated or dark adapted leaves. Photosynth Res 31: 11–19
Richter M, Goss R, Wagner B and Holzwarth AR (1999) Characterization of the fast and slow reversible components of non-photochemical quenching in isolated pea thylakoids by picosecond time resolved chlorophyll fluorescence analysis. Biochemistry 38: 12718–12726
Ruban AV, Young AJ and Horton P (1994) Modulation of chlorophyll fluorescence quenching in isolated light harvesting complex of Photosystem II. Biochim Biophys Acta 1186: 123–127
Ruban AV, Young AJ and Horton P (1996) Dynamic properties of the minor chlorophyll a/b binding proteins of Photosystem II, an in vitro model for photoprotective energy dissipation in the photosynthetic membrane of green plants. Biochemistry 35: 674–678
Ruban AV, Calkoen F, Kwa SLS, Van Grondelle R, Horton P and Dekker J (1997a) Characterisation of LHC II in the aggregated state by linear and circular dichroism spectroscopy. Biochim Biophys Acta 1321: 61–70
Ruban AV, Phillip D, Young AJ and Horton P (1997b)Carotenoiddependent oligomerization of the major chlorophyll a/b lightharvesting complex of Photosystem-II of plants. Biochemistry 36: 7855–7859
Ruban AV, Phillip D, Young AJ and Horton P (1998) Excited-state energy level does not determine the differential effect of violaxanthin and zeaxanthin on chlorophyll fluorescence quenching in isolated light-harvesting complex of Photosystem II. Photochem Photobiol 68: 829–834
Simidjiev I, Barzda V, Mustárdy L and Garab G (1997) Isolation of lamellar aggregates of the light-harvesting chlorophyll a/b protein complex of Photosystem II with long-range chiral order and structural flexibility. Anal Biochem 250: 169–175
Staehelin LA and Arntzen CJ (1983) Regulation of chloroplast membrane function: Protein phosphorylation changes the spatial organization of membrane components. J Cell Biol 97: 1327–1337
Van Metter RL (1977) Excitation energy transfer in the lightharvesting chlorophyll a/b protein. Biochim Biophys Acta 462: 642–658
Wagner B, Goss R, Richter M and Holzwarth AR (1996) Picosecond time-resolved study on the nature of high-energy state quenching in isolated pea thylakoids. Different localization of zeaxanthin dependent and independent quenching mechanisms. J Photochem Photobiol B Biol 36: 339–350
Wilhelm C and Duval JC (1990) Fluorescence induction kinetics as a tool to detect a chlororespiratory activity in the prasinophycean alga Mantoniella squamata. Biochim Biophys Acta 1016: 197–202
Zer H, Vink M, Keren N, Dilly-Hartwig HG, Paulsen H, Herrmann RG, Andersson B and Ohad I (1999) Regulation of thylakoid protein phosphorylation at the substrate level: Reversible lightinduced conformational changes expose the phosphorylation site of the light-harvesting complex II. Proc Natl Acad Sci USA 96: 8277–8282
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goss, R., Garab, G. Non-photochemical chlorophyll fluorescence quenching and structural rearrangements induced by low pH in intact cells of Chlorella fusca (Chlorophyceae) and Mantoniella squamata (Prasinophyceae) . Photosynthesis Research 67, 185–197 (2001). https://doi.org/10.1023/A:1010681511105
Issue Date:
DOI: https://doi.org/10.1023/A:1010681511105