Abstract
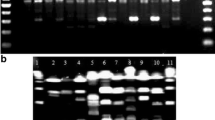
Eight strains of Taylorella equigenitalis were identified by a polymerase chain reaction using a primer pair specific to the 16S rDNA of T. equigenitalis. These eight strains were chosen because they had previously been shown to represent eight distinct genotypes by pulsed-field gel electrophoresis analysis after separate digestion of the genomic DNA with ApaI or NotI. The eight strains could be classified into six or seven types by random amplified polymorphic DNA analysis using different kinds of primers. Amplified rDNA restriction analysis after separate digestion with five restriction enzymes, including AluI and MboI, of the 1500 bp fragments of rDNA amplified by polymerase chain reaction did not discriminate the genomic variations among the eight strains of T. equigenitalis. Thus, pulsed-field gel electrophoresis was shown to discriminate these eight organisms better than random amplified polymorphic DNA analysis, while amplified rDNA restriction analysis was found to be unsuitable for subtyping T. equigenitalis.
Similar content being viewed by others
REFERENCES
Akopyanz, N., Bukanov, N.O., Westblom, T.U., Kresovich, S. and Berg, D.E., 1992. DNA diversity among clinical isolates of Helicobacter pylori detected by PCR-based RAPD fingerprinting. Nucleic Acids Research, 20, 5137–5142
Bleumink-Pluym, N., Ter Laak, E.A. and Van Der Zeijst, B.A.M., 1990. Epidemiologic study of Taylorella equigenitalis strains by field inversion gel electrophoresis of genomic restriction endonuclease fragments. Journal of Clinical Microbiology, 28, 2012–2016
Chetoui, H., Melin, P., Struelens, M.J., Delhalle, E., Nigo, M.M., De Ryck, R. and De Mol, P., 1997. Comparison of biotyping, ribotyping, and pulsed-field gel electrophoresis for investigation of a common-source outbreak of Burkholderia pickettii bacteremia. Journal of Clinical Microbiology, 35, 1398–1403
Crowhurst, R.C., 1977. Genital infection in mares. The Veterinary Record, 100, 476
Endtz, H.P., Vliegenthart, J.S., Vandamme, P., Weverink, H.W., Van Den Braak, N.P., Verbrugh, H.A. and Van Belkum, A., 1997. Genotypic diversity of Campylobacter lari isolated from mussels and oysters in the Netherlands. International Journal of Food Microbiology, 34, 79–88
Gomez-Lus, P., Fields, B.S., Benson, R.F., Martin, W.T., O'Connor, S.P. and Black, C.M., 1993. Comparison of arbitrarily primed polymerase chain reaction, ribotyping, and monoclonal antibody analysis for subtyping Legionella pneumophila serogroup I. Journal of Clinical Microbiology, 31, 1940–1942
Liebisch, B. and Schwarz, S., 1996. Evaluation and comparison of molecular techniques for epidemiological typing of Salmonella enterica subsp. enterica serovar dublin. Journal of Clinical Microbiology, 34, 641–646
Matsuda, M., Miyazawa, T., Ishida, Y. and Moore, J.E., 1997. Profiles of fragments after pulsed-field gel electrophoresis of cleaved genomic DNA from strains of Taylorella equigenitalis isolated from horses in Norway. Microbiological Research, 152, 217–220
Matsuda, M., Miyazawa, T., Moore, J.E., Buckley, T.C. and Thomas, L.A., 1998. Molecular genotyping by pulsed-field gel electrophoresis of restricted genomic DNA of strains of Taylorella equigenitalis isolated in Ireland and in the United States. Veterinary Research Communications, 22, 217–224
Matsuda, M., Kagawa, S., Sakamoto, Y., Miyajima, M., Barton, M. and Moore, J.E., 2000. Detection of heterogeneous genotypes among Australian strains of Taylorella equigenitalis. Australian Veterinary Journal, 78, 56–57
Miserez, R., Frey, J., Krawinkler, M. and Nicolet, J., 1996. Identifikation und diagnostik von Taylorella equigenitalis mittels einer DNA-amplifikationsmethode (PCR). Schweizer Archiv fur Tierheilkunde, 138, 115–120
Miyazawa, T., Matsuda, M., Isayama, Y., Samata, T., Ishida, Y., Ogawa, S., Takei, K., Honda, M. and Kamada, M., 1995. Genotyping of isolates of Taylorella equigenitalis from thoroughbred brood mares in Japan. Veterinary Research Communications, 19, 265–271
Old, D.C., Rankin, S.C. and Crichton, P.B., 1999. Assessment of strain relatedness among Salmonella serotypes Salinatis, Duisberg, Sandiego by biotyping, ribotyping, IS200 fingerprinting, and pulsed-field gel electrophoresis. Journal of Clinical Microbiology, 37, 1687–1692
Rafferty, M.E., Baltch, A.L., Smith, R.P., Bopp, L.H., Rheal, C., Tenover, F.C., Killgore, G.E., Lyerly, D.M., Wilkins, T.D., Schoonmaker, D.J., Hannett, G.E. and Shayegani, M., 1998. Comparison of restriction enzyme analysis, arbitrarily primed PCR, and protein profile analysis typing for epidemiologic investigation of an ongoing Clostridium difficile outbreak. Journal of Clinical Microbiology, 36, 2957–2963
Sambrook, J., Fritsch, E.F. and Maniatis, T., 1989. Molecular Cloning: A Laboratory Manual, 2nd edn, (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, USA)
Savor, C., Pfaller, M.A., Kruszynski, J.A., Hollis, R.J., Noskin, G.A. and Peterson, L.R., 1998. Comparison of genomic methods for differentiating strains of Enterococcus faecium: assessment using clinical epidemiologic data. Journal of Clinical Microbiology, 36, 3327–3331
Taylor, C.E.D., Rosenthal, R.O., Brown, D.F.J., Lapage, S.P., Hill, L.R. and Legros, R.M., 1978. The causative organism of contagious equine metritis 1977; proposal for a new species to be known as Haemophilus equigenitalis. Equine Veterinary Journal, 10, 136–144
Ter Laak, E.A., Fennema, G. and Jaartsveld, F.H.J., 1989. Contagious equine metritis in the Netherlands. Tijdschrift voor Diergeneeskdunde, 114, 189–201
Timoney, P.J. and Powell, D.G., 1982. Isolation of the contagious equine metritis organism from colts and fillies in the United Kingdom and Ireland. The Veterinary Record, 111, 478–482
Timoney, P.J. and Strickland, K.L., 1982. CEM in the Republic of Ireland. The Veterinary Record, 111, 400–401
Van Belkum, A., Bax, R., Peerbooms, P., Goessens, W.H.F., Van Leeuwen, N. and Quint, W.G.V., 1993. Comparison of phage typing and DNA fingerprinting by polymerase chain reaction for discrimination of methicillin-resistant Staphylococcus aureus strains. Journal of Clinical Microbiology, 31, 798–803
Van Der Zee, A., Verbakel, H., Van Zon J.-C., Frenay, I., Van Belkum, A., Peeters, M., Buiting, A. and Bergmans, A., 1999. Molecular genotyping of Staphylococcus aureus strains: comparison of repetitive element sequence-based PCR with various typing methods and isolation of a novel epidemicity marker. Journal of Clinical Microbiology, 37, 342–349
Wada, S., Matsuda, M., Kikuchi, M., Kodama, T., Takei, I., Ogawa, S., Takahashi, S., Shingaki, M. and Itoh, T., 1994. Genome DNA analysis and genotyping of clinical isolates of Helicobacter pylori. Cytobios, 80, 109–116
Weisburg, W.G., Barns, S.M., Pelletier, D.A. and Lane, D.J., 1991. 16S ribosomal DNA amplification for phylogenetic study. Journal of Bacteriology, 173, 697–703
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kagawa, S., Moore, J., Murayama, O. et al. Comparison of the Value of Pulsed-field Gel Electrophoresis, Random Amplified Polymorphic DNA and Amplified rDNA Restriction Analysis for Subtyping Taylorella equigenitalis. Vet Res Commun 25, 261–269 (2001). https://doi.org/10.1023/A:1010674524428
Issue Date:
DOI: https://doi.org/10.1023/A:1010674524428