Abstract
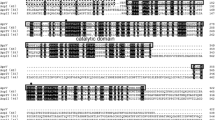
The gene, vapK, encoding an extracellular alkaline protease, VapK, in Vibrio metschnikovii strain RH530 was cloned and expressed in Escherichia coli. The nucleotide sequence of the vapK revealed a single open reading frame of 1266 bp encoding 422 amino acids. The biochemical characteristics of the expressed protease were indistinguishable from those of VapK from the wild type strain. The recombinant VapK showed resistant activity to surfactants such as sodium α-olefin sulfonate, polyoxyethylene oxide, SDS and urea. The surfactant, sodium alkylbenzenesulfonate, strongly inhibited the activity of re-VapK. However in the presence of POE or Na2SO4, its activity was completely restored. By N-terminal sequencing of VapK, the cleavage site for the mature protease could be identified. The cleavage site was Ala140 and the calculated molecular mass of the mature enzyme was 29 609 Da, corresponding to the purified VapK. The active sites and putative calcium binding sites of recombinant VapK (re-VapK) were highly conserved when compared to subtilisin Carlsberg.
Similar content being viewed by others
References
Banerjee UC, Sani RK, Azmi W, Soni R (1999) Thermostable alkaline protease from Bacillus brevis and its characterization as a laundry detergent additive. Proc. Biochem. 35: 213–219.
Birnboim HC, Doly J (1979) A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucl. Acids Res. 7: 1513–1523.
Deane SM, Robb FT, Woods DR (1987) Production and activation of a SDS-resistant alkaline serine exoprotease of Vibrio alginolyticus. J. Gen. Microbiol. 133: 391–398.
Gunkel FA, Gassen HG (1989) Proteinase K from Tritirachium album Limber. Eur. J. Biochem. 179: 185–194.
Hemachander C, Puvanakrishnan R (2000) Lipase from Ralstonia pickettii as an additive in laundry detergent formulations. Proc. Biochem. 35: 809–814.
Kaneto R, Koyama N, Tsai YC, Juang RY, Yoda K, Yamasaki M (1989) Molecular cloning of the structural gene for alkaline elastase YaB, a new subtilisin produced by an alkalophilic Bacillus strain. J. Bacteriol. 171: 5232–5236.
Ko H, Jang WH, Kim EK, Lee HH, Park KD, Chung JH, Yoo OJ (1996) Enhancement of thermostability and catalytic efficiency of AprP, an alkaline protease from Pseudomonas sp., by the introduction of a disulfide bond. Biochem. Biophys. Res. Commun. 221: 631–635.
Kwon YT, Kim JO, Moon SY, Lee HH, Rho HM (1994) Extracellular alkaline proteases from alkalophilic Vibrio metschnikovii strain RH530. Biotechnol. Lett. 16: 413–418.
Kwon YT, Kim JO, Moon SY, Yoo YD, Rho HM (1995) Cloning and charactrization of the gene encoding an extracellular serine protease from Vibrio metschnikovii strain RH530. Gene 152: 59–63.
Kwon YT, Moon SY, Kim JO, Kho YH, Rho HM (1992) Characterization of extracellular proteases from alkalophilic Vibrio sp. strain RH530. Kor. J. Microbiol. 30: 501–506.
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Mamur J (1961) A procedure for the isolation of deoxyribonucleic acid from microorganisms. J. Mol. Biol. 3: 208–218.
Matsudaira P (1987) Sequence from picomole quantities of proteins electroblotted onto polyvinylidene difluoride membranes. J. Biol. Chem. 262: 10035–10038
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: A Laboratory Manual, 2nd edn. New York: Cold Spring Harbor Laboratory Press.
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain terminating inhibitor. Proc. Natl. Acad. Sci. USA. 74: 5463–5467.
Smith ES, De Lange RJ, Evans WH, Landon M, Markland FS (1968) Subtilisin Carlsberg. V. The complete sequence: comparison with subtilisin BPN: evolutionary relationships. J. Biol. Chem. 243: 2184–2191.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, S.S., Kim, H.J., Shin, Y.U. et al. Molecular characterization of gene encoding an extracellular alkaline protease in Vibrio metschnikovii. Biotechnology Letters 23, 1175–1182 (2001). https://doi.org/10.1023/A:1010525428239
Issue Date:
DOI: https://doi.org/10.1023/A:1010525428239