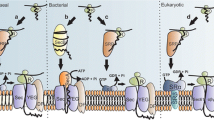
Abstract
The signal recognition particle (SRP) is a conserved ribonucleoprotein complex that binds to targeting sequences in nascent secretory and membrane proteins. The SRP guides these proteins to the cytoplasmic membrane in prokaryotes and the endoplasmic reticulum membrane in eukaryotes via an interaction with its cognate receptor. The E. coli SRP is relatively small and is currently used as a model for fundamental and applied studies on translation-linked protein targeting. In this review recent advances in our understanding of the structure and function of the E. coli SRP and its receptor are discussed. In particular, the interplay between the SRP pathway and other targeting routes, the role of guanine nucleotides in cycling of the SRP and the substrate specificity of the SRP are highlighted
Similar content being viewed by others
References
Altamura N, Capitanio N, Bonnefoy N, Papa S & Dujardin G (1996) The Saccharomyces cerevisiae OXAl gene is required for the correct assembly of cytochrome c oxidase and oligomycin-sensitive ATP synthase. FEBS Lett. 382: 111-115
Bacher G, Lütcke H, Jungnickel B, Rapoport TA & Dobberstein B (1996) Regulation by the ribosome of the GTPase of the signalrecognition particle during protein targeting. Nature 381: 248-251
Bassford P, Beckwith J, Ito K, Kumamoto C, Mizushima S, Oliver D, Randall L, Silhavy T, Tai PC & Wickner B (1991) The primary pathway of protein export in E. coli. Cell 65: 367-368
Beckmann R, Bubeck D, Grassucci R, Penczek P, Verschoor A, Blobel G & Frank J (1997) Alignment of conduits for the nascent polypeptide chain in the Ribosome-Sec6l complex. Science 278: 2l23-2126
Belin D, Bost S, Vassalli JD & Strub K (1996) A two-step recognition of signal sequences determines the translocation efficiency of proteins. EMBO J. 15: 468-478
Berks BC (1996) A common export pathway for proteins binding complex redox cofactors? Mol. Microbiol. 22: 393-404
Bernstein HD, Poritz MA, Strub K, Hoben PJ, Brenner S & Walter P (1989) Model for signal sequence recognition from amino-acid sequence of 54K subunit of signal recognition particle. Nature 340: 482-486
Bernstein HD, Zopf D, Freymann DM & Walter P (1993) Functional substitution of the signal recognition particle 54-kDa subunit by its Escherichia coli homolog. Proc. Natl. Acad. Sci. USA 90: 5229-5233
Bernstein HD (1998) Protein targeting: Getting into the groove. Curr. Biol. 8: R715-R718
Blattner FR, Plunkett G, Bloch CA, Perna NT, Burland V, Riley M, ColladoVides J, Glasner JD, Rode CK, Mayhew GF, Gregor J, Davis NW, Kirkpatrick HA, Goeden MA, Rose DJ, Mau B & Shao Y (1997) The complete genome sequence of Escherichia coli K-12. Science 277: 1453
Bourne HR, Sanders DA & McCormick F (1990) The GTPase superfamily: a conserved switch for diverse cell functions. Nature 348: 125-132
Brodsky JL (1998) Translocation of proteins across the endoplasmic reticulum membrane. Intern. Rev. Cytol 1.78: 277-327
Bunai K, Yamada K, Hayashi K, Nakamura K & Yamane K (1998) Enhancing effect of Bacillus subtilis Ffh, a homologue of the SRP54 subunit of the mammalian signal recognition particle, on the binding of SecA to precursors of secretory proteins in vitro. J. Biochem. 125: 151-159
Carson MJ, Barondess I & Beckwith J (1991) The FtsQ protein of Escherichia coli: membrane topology, abundance and cell division phenotypes due to overproduction and insertion mutations. J. Bacteriol. 173: 2187-2195
Connolly T & Gilmore R (1989) The signal recognition particle receptor mediates the GTP-dependent displacement of SRP from the signal sequence of the nascent polypeptide. Cell 57: 599-610
Connolly T & Gilmore R (1993) GTP hydrolysis by complexes of the signal recognition particle and the signal recognition particle receptor. J. Cell Biol. 123: 799-807
Connolly T, Rapiejko PJ & Gilmore R (1991) Requirement of GTP hydrolysis for dissociation of the signal recognition particle from its receptor. Science 252: 1171-1173
Dalbey RE (1991) Leader peptidase. Mol. Microbiol. 5: 2855-2860
Dalbey RE, Kuhn A & von Heijne G (1995) Directionality in protein translocation across membranes: The N-tail phenomenon. Trends Cell Biol. 5: 380-383
Dalbey RE & Robinson C (1999) Protein translocation into and across the bacterial plasma membrane and the plant thylakoid membrane. TIBS 24: 17-22
De Gier J-WL, Mansournia P, Valent QA, Phillips GJ, Luirink J & von Heijne G (1996) Assembly of a cytoplasmic membrane protein in Escherichia coli is dependent on the signal recognition particle. FEBS Lett. 399: 307-309
De Leeuw E, Poland D, Mol O, Sinning I, ten Hagen-Jongman CM, Oudega B & Luirink J (1997) Membrane association of FtsY, the E-coli SRP receptor. FEBS Lett. 416: 225-229
Diamond DL & Randall LL (1997) Kinetic partitioning-Poising SecB to favor association with a rapidly folding ligand. J. Biol. Chem. 272: 28994-28998
Doud SK, Chou MM & Kendall DA (1993) Titration of protein transport activity by incremental changes in signal peptide hydrophobicity. Biochem. 32: 1251-1256
Driessen AJM (1993) SecA, the peripheral pubunit of the Escherichia-coli precursor protein translocase, is functional as a dimer. Biochem. 32: 1319O-13197
Driessen AJM, Fekkes P & Van der Wolk JPW (1998) The Sec system. Curr. Opin. Microbiol. 1: 216-222
Duong F & Wickner W (1997) The SecDFyajC domain of preprotein translocase controls preprotein movement by regulating SecA membrane cycling. EMBO J. 16: 4871-4879
Economou A, Pogliano JA, Beckwith J, Oliver DB & Wickner W (1995) SecA membrane cycling at SecYEG is driven by distinct ATP binding and hydrolysis events and is regulated by SecD and SecF. Cell 83: 1171-1181
Economou A & Wickner W (1994) SecA promotes preprotein translocation by undergoing ATP-driven cycles of membrane insertion and deinsertion. Cell 78: 835-843
Fekkes P, Van der Does C & Driessen AJM (1997) The molecular chaperone SecB is released from the carboxy-terminus of SecA during initiation of precursor protein translocation. EMBO J. 16: 6105-6113
Freymann DM, Keenan RJ, Stroud RM & Walter P (1997) Structure of the conserved GTPase domain of the signal recognition particle. Nature 385: 361-364
Gill DR & Salmond GPC (1990) The identification of the Escherichia coli ftsY gene product: an unusual protein. Mol. Microbiol 4: 575-583
Hamman BD, Hendershot LM & Johnson AE (1998) BiP maintains the permeability barrier of the ER membrane by sealing the lumenal end of the translocon pore before and early in translocation. Cell 92: 747-758
Hatsuzawa K, Tagaya M & Mizushima S (1997) Hydrophobic region of signal peptides is a determinant for SRP recognition and protein translocation across the ER membrane. J. Biochem. 121: 270-277
Hauser S, Bacher G, Dobberstein B & Lütcke H (1995) A complex of the signal sequence binding protein and the SRP RNA promotes translocation of nascent proteins. EMBO J. 14: 5485-5493
He SC & Fox TD (1997) Membrane translocation of mitochondrially coded Cox2p: Distinct requirements for export of N and C termini and dependence on the conserved protein Oxalp. Mol. Biol. Cell 8: 1449-1460
Hell K, Herrmann J, Pratje E, Neupert W & Stuart RA (1997) Oxalp mediates the export of the N-and C-termini of pCoxII from the mitochondrial matrix to the intermembrane space. FEBS Lett. 418: 367-370
Hell K, Herrmann J, Pratje E, Neupert W & Stuart RA (1998) Oxalp, an essential component of the N-tail protein export machinery in mitochondria. Proc. Natl. Acad. Sci. USA 95: 2250-2255
High S, Henry R, Mould RM, Valent QA, Meacock S, Cline K, Gray JC & Luirink J (1997) Chloroplast SRP54 interacts with a specific subset of thylakoid precursor proteins. J. Biol. Chem. 272: 1162-11628
Johnson AE (1997) Protein translocation at the ER membrane: A complex process becomes more so. Trends Cell Biol. 7: 90-95
Jungnickel B & Rapoport TA (1995) A posttargeting signal sequence recognition event in the endoplasmic reticulum membrane. Cell 82: 261-270
Keenan RJ, Freymann DM, Walter P & Stroud RM (1998) Crystal structure of the signal sequence binding subunit of the signal recognition particle. Cell 94: 181-191
Kumamoto CA & Beckwith J (1985) Evidence for specificity at an early step in protein export in Escherichia coli. J. Bacteriol. 163: 267-274
Kumamoto CA & Francetic O (1993) Highly selective binding of nascent polypeptides by an Escherichia coli chaperone protein in vivo. J. Bacteriol. 175: 2184-2188
Kusters R, De Vrije T, Breukink E & De Kruijff B (1989) SeuB stabilizes a translocation-competent state of purified prePhoE protein. J. Biol. Chem. 264: 20827-20830
Kusters R, Lentzen G, Eppens E, Van Geel A, Van der Weijden CC, Wintermeyer W & Luirink J (1995) The functioning of the SRP receptor FtsY in protein-targeting in E-coli correlated with its ability to bind and hydrolyse GTP. FEBS Lett. 372: 253-258
Lecker S, Lill R, Ziegelhoffer T, Georgopoulos C, Bassford PJ, Kumamoto CA & Wickner W (1989) Three pure chaperone proteins of Escherichia coli-SecB, trigger factor and GroEL-form soluble complexes with precursor proteins in vitro. EMBO J. 8: 2703-2709
Liao SR, Lin JL, Do H & Johnson AE (1997) Both lumenal and cytosolic gating of the aqueous ER translocon pore are regulated from inside the ribosome during membrane protein integration. Cell 90: 31-41
Luirink J, High S, Wood H, Giner A, Tollervey D & Dobberstein B (1992) Signal sequence recognition by an Escherichia coli ribonucleoprotein complex. Nature 359: 741-743
Luirink J, Ten Hagen-Jongman CM, Van der Weijden CC, Oudega B, High S, Dobberstein B & Kusters R (1994) An alternative protein targeting pathway in Escherichia coli: Studies on the role of FtsY. EMBO J. 13: 2289-2296
Lütcke H (1995) Signal recognition particle (SRP), a ubiquitous initiator of protein translocation. Eur. J. Biochem. 228: 531-550
MacFarlane J & Müller M (1995) The functional integration of a polytopic membrane protein of Escherichia coli is dependent on the bacterial signal-recognition particle. Eur. J. Biochem. 233: 766-771
Miller JD, Bernstein HD & Walter P (1994) Interaction of E. coli Ffh/4.5S ribonucleoprotein and FtsY mimics that of mammalian signal recognition particle and its receptor. Nature 367: 657-659
Miller JD, Wilhelm H, Gierasch L, Gilmore R & Walter P (1993) GTP binding and hydrolysis by the signal recognition particle during initiation of protein translocation. Nature 366: 351-354
Mitsopoulos C, Hashemzadeh-Bonehi L & Broome-Smith JK (1997) N-tail translocation of mature beta-lactamase across the Escherichia coli cytoplasmic membrane. FEBS Lett. 419: 18-22
Montoya G, Svensson C, Luirink J & Sinning I (1997) Crystal structure of the NG domain from the signal-recognition particle receptor FtsY. Nature 385: 365-368
Newitt JA & Bernstein HD (1997) The N-domain of the signal recognition particle 54-kDa subunit promotes efficient signal sequence binding. Eur. J. Biochem. 245: 720-729
Ng DTW, Brown JD & Walter P (1996) Signal sequences specify the targeting route to the endoplasmic reticulum membrane. J. Cell Biol. 134: 269-278
Oh DB, Yi GS, Chi SW & Kim H (1996) Structure of a methioninerich segment of Escherichia coli Ffh protein. FEBS Lett. 395: 160-164
Phillips GJ & Silhavy TJ (1992) The E. coli ffh gene is necessary for viability and efficient protein export. Nature 359: 744-746
Poritz MA, Bernstein HD, Strub K, Zopf D, Wilhelm H & Walter P (1990) An E. coli ribonucleoprotein containing 4.5S RNA resembles mammalian signal recognition particle. Science 250: 1111-1117
Poritz MA, Strub K & Walter P (1988) Human SRP RNA and E. coli 4.5S RNA contain a highly homologous structural domain. Cell 55: 4-6
Powers T. & Walter P (1995) Reciprocal stimulation of GTP hydrolysis by two directly interacting GTPases. Science 269: 1422-1424
Powers T & Walter P (1997) Co-translational protein targeting catalyzed by the Escherichia coli signal recognition particle and its receptor. EMBO J. 16: 4880-4886
Powers T & Walter P (1997) Protein synthesis-A ribosome at the end of the tunnel. Science 278: 2072-2073
Pugsley AP (1993) The complete general secretory pathway in gram-negative bacteria. Microbiol. Rev. 57: 50-108
Qi H-Y & Bernstein HD (1999) SecA is required for the insertion of inner membrane proteins targeted by the Escherichia coli signal recognition particle. J. Biol. Chem. 274: 8993-8997
Rapiejko PJ & Gilmore R (1994) Signal sequence recognition and targeting of ribosomes to the endoplasmic reticulum by the signal recognition particle do not require GTP. Mol. Biol. Cell 5: 887-897
Rapiejko PJ & Gilmore R (1997) Empty site forms of the SRP54 and SR alpha GTPases mediate targeting of ribosome nascent chain complexes to the endoplasmic reticulum. Cell 89: 703-713
Rapoport T (1991) A bacterium catches up. Nature 349: 107-108
Rapoport TA, Jungnickel B & Kutay U (1996) Protein transport across the eukaryotic endoplasmic reticulum and bacterial inner membranes. Ann. Rev. Biochem. 65: 271-303
Ribes V, Römisch K, Giner A, Dobberstein B & Tollervey D (1990) E. coli 4.5S RNA is part of a ribonucleoprotein particle that has properties related to signal recognition particle. Cell 63: 591-600
Römisch K, Webb J, Herz J, Prehn S, Frank R, Vingron M & Dobberstein B (1989) Homology of 54K protein of signal recognition particle, docking protein and two E. coli proteins with putative GTP-binding domains. Nature 340: 478-482
Römisch K, Webb J, Lingelbach K, Gausepohl H & Dobberstein B (1990) The 54-kD protein of signal recognition particle contains a methionine-rich RNA binding domain. J. Cell Biol. 111: 1793-1802
Sambrook J, Fritsch EF & Maniatisi T (1989) Molecular cloning: a laboratory manual. 2nd edition, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Samuelsson T, Olsson M, Wikström PM & Johansson BR (1995) The GTPase activity of the Escherichia coli Ffh protein is important for normal growth. BBA-Mol. Cell Res. 1267: 83-91
Santini CL, Ize B, Chanal A, Müller M, Giordano G & Wu LF (1998) A novel Sec-independent periplasmic protein translocation pathway in Escherichia coli. EMBO J. 17: 101-112
Scotti PA, Valent QA, Manting EH, Urbanus ML, Driessen AJM, Oudega B & Luirink J (1999) SecA is not required for signal recognition particle-mediated targeting and initial membrane insertion of a nascent inner membrane protein. J. Biol. Chem. 274: 29883-29888
Shimizu H, Nishiyama K & Tokuda H (1997) Expression of gpsA encoding biosynthetic sn-glycerol 3-phosphate dehydrogenase suppresses both the LB-phenotype of a SecB null mutant and the cold-sensitive phenotype of a SecG null mutant. Mol. Microbiol. 26: 1013-1021
Siegel V & Walter P (1985) Elongation arrest is not a prerequisite for secretory protein translocation across the microsomal membrane. J. Cell Biol. 100: 1913-1921
Siegel V & Walter P (1988) Functional dissection of the signal recognition particle. TIBS 13: 314-315
Stephens C (1998) Protein secretion: Getting folded proteins across membranes. Curr. Biol. 8: R578-R581
Ulbrandt ND, Newitt JA & Bernstein HD (1997) The E. coli signal recognition particle is required for the insertion of a subset of inner membrane proteins. Cell 88: 187-196
Valent QA, Kendall DA, High S, Kusters R, Oudega B & Luirink J (1995) Early events in preprotein recognition in Escherichia coli interaction of SRP and trigger factor with nascent polypeptides. EMBO J. 14: 5494-5505
Valent QA, De Gier J-WL, von Heijne G, Kendall DA, Ten Hagen-Jongman CM, Oudega B & Luirink J (1997) Nascent membrane and presecretory proteins synthesized in Escherichia coli associate with signal recognition particle and trigger factor. Mol. Microbiol. 25: 53-64
Valent QA, Scotti PA, High S, De Gier J-WL, von Heijne G, Lentzen G, Wintermeyer W, Oudega B & Luirink J (1998) The E. coli SRP and SecB targeting pathways converge at the translocon. EMBO J. 17: 2504-2512
Van der Wolk JPW, De Wit JG & Driessen AJM (1997) The catalytic cycle of the Escherichia coli SecA ATPase comprises two distinct preprotein translocation events. EMBO J. 16: 7297-7304
von Heijne, G & Segrest, JP (1987) The leader peptides from bacteriorhodopsin and halorhodopsin are potential membrane-spanning amphipathic helices, FEBS Lett., 213, 1716-1719
Walter P & Johnson AE (1994) Signal sequence recognition and protein targeting to the endoplasmic reticulum membrane. Ann. Rev. Cell Biol: 87-119
Weiner JH, Bilous PT, Shaw GM, Lubitz SP, Frost L, Thomas GH, Cole JA & Turner RJ (1998) A novel and ubiquitous system for membrane targeting and secretion of cofactor-containing proteins. Cell 93: 93-101
Wikström PM & Björk GR (1988) Noncoordinate translation-level regulation of ribosomal and nonribosomal protein genes in the Escherichia coli trmD operon. J. Bacteriol. 170: 3025-3031
Wilkinson BM, Regnacq M & Stirling CJ (1997) Protein translocation across the membrane of the endoplasmic reticulum. J. Mem. Biol. 155: 189-197
Wood H, Luirink J & Tollervey D (1992) Evolutionary conserved nucleotides within the E. coli 4.5S RNA are required for association with P48 and for optimal function in vivo. Nucl. Acids Res. 20: 5919-5925
Young JC, Ursini J, Legate KR, Miller JD, Walter P & Andrews DW (1995) An amino-terminal domain containing hydrophobic and hydrophilic sequences binds the signal recognition particle receptor α subunit to the β subunit on the endoplasmic reticulum membrane. J. Biol. Chem. 270: 15650-15657
Zelazny A, Seluanov A, Cooper A & Bibi E (1997) The NG domain of the prokaryotic signal recognition particle receptor, FtsY, is fully functional when fused to an unrelated integral membrane polypeptide. Proc. Natl. Acad. Sci. USA 94: 6025-6029
Zopf D, Bernstein HD, Johnson AE & Walter P (1990) The methionine-rich domain of the 54 kd protein subunit of the signal recognition particle contains an RNA binding site and can be crosslinked to a signal sequence. EMBO J. 9: 4511-4517
Zopf D, Bernstein RD & Walter P (1993) GTPase domain of the 54-kD subunit of the mammalian signal recognition particle is required for protein translocation but not for signal sequence binding. J. Cell Biol. 120: 1113-1121
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valent, Q.A. Signal recognition particle mediated protein targeting in Escherichia coli. Antonie Van Leeuwenhoek 79, 17–31 (2001). https://doi.org/10.1023/A:1010256109582
Issue Date:
DOI: https://doi.org/10.1023/A:1010256109582