Abstract
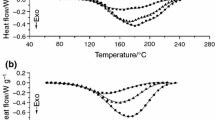
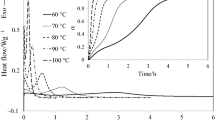
The curing characteristics of adicyandiamide-cured epoxy system under the influence of solvents in a closed environment were studied by means of isothermal differential scanning calorimetry (DSC) and Fourier transform infrared spectroscopy (FTIR). The DSC analyses revealed that the presence of solvent results in decreases in the curing exotherm, the initial curing rate, the glass transition temperature, the reaction rate and the reaction order of the epoxy resin. The greatest decreases were caused by the solvent with the highest boiling temperature. A change in temperature-dependent curing route due to the heat absorbed during solvent evaporation is responsible for the difference. The FTIR analyses confirmed that the composition of the cured resin is affected by the solvent, the curing temperature and the specimen configuration. As compared with those obtained from open systems, specimens produced in a closed environment have an enhanced curing exotherm, initial curing rate, glass transition temperature, reaction rate and reaction order because of the retention of volatile catalytic by-products.
Similar content being viewed by others
References
E. Sacher, Polymer, 14 (1973) 91.
S. Muroi, H. Ishimura and M. Outsuka, J. Appl. Polym. Sci., 32 (1986) 5095.
R. L. Miller and M. A. Oebser, Thermochim. Acta, 36 (1980) 121.
P. N. Son and C. D. Weber, J. Appl. Polym. Sci., 17 (1973) 1305.
J. M. Barton and D. C. Greenfield, Brit. Polym. J., 18 (1986) 196.
Y. G. Lin, H. Sautereau and J. P. Pascault, J. Polym. Sci., Part A, 24 (1986) 2171.
T. Guthner and B. Hammer, J. Appl. Polym. Sci., 50 (1993) 1453.
M.-F. Grenier-Loustalot, M.-P. Bente and P. Grenier, Eur. Polym. J., 27 (1991) 1201.
M. D. Gilbert, N. S. Schneider and W. J. MacKnight, Macromolecules, 24 (1991) 360.
M. Opresnik, A. Sebenik, M. Zigon and U. Osredkar, Thermochim. Acta, 178 (1991) 127.
S. G. Hong and T. C. Wang, Thermochim. Acta, 237 (1994) 305.
S. G. Hong and J. J. Lin, J. Appl. Polym. Sci., 59 (1996) 1597.
R. O. Cater III, R. A. Dickie, J. W. Holubka and N. E. Lindsay, Ind. Eng. Chem. Res., 28 (1989) 48.
R. O. Cater III, R. A. Dickie and J. W. Holubka, Polym. Mat. Sci. Eng., 58 (1988) 55.
W. Brockmann, O. D. Hennemann, H. Kollek and C. Matz, Int. J. Adhes. Adhesives, 6 (1986) 115.
F. J. Boerio and P. P. Hong, Mat. Sci. Eng., A126 (1990) 245.
G. L. Hagnauer and D. A. Dunn, J. Appl. Polym. Sci., 26 (1981) 1837.
H. Inoue, H. Fukke, M. Katsumoto and K. Konno, J. Colloid Interface Sci., 138 (1990) 92.
M. Shimbo, M. Ochi and K. Arai, J. Coating Technol., 57 (1985) 93.
S. G. Croll, J. Coating Technol., 51 (1979) 49.
S. G. Croll, J. Coating Technol., 53 (1981) 85.
S. G. Hong and C. S. Wu, Thermochim. Acta, in press, 1998.
R. A. Fava, Polymer, 9 (1968) 137.
G. L. Hagnauer and D. A. Dunn, J. Appl. Polym. Sci., 26 (1981) 1837.
B. C. Ennis, R. G. Davidson, P. J. Pearce and C. E. M. Morris, J. Adhesion, 37 (1992) 131.
T. F. Saunders, M. F. Levy and J. F. Serino, J. Polym. Sci., Part A-1, 5 (1967) 1609.
P. Peyser and W. D. Bascom, J. Appl. Polym. Sci., 21 (1977) 2359.
Y. G. Lin, H. Sautereau and J. P. Pascault, J. Polym. Sci., Part A, 24 (1986) 2171.
M. D. Gilbert, N. S. Schneider and W. J. MacKnight, Macromolecules, 24 (1991) 360.
P. Eyerer, J. Appl. Polym. Sci., 15 (1971) 3067.
H. H. Levine, Polym. Preprint, 41 (1964) 8.
R. O. Cater III, R. A. Dickie, J. W. Holubka and N. E. Lindsay, Ind. Eng. Chem. Res., 28 (1989) 48.
W. J. Jones and W. J. Orville-Thomas, Trans. Faraday Soc., 55 (1959) 193.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hong, SG., Wu, CS. DSC and FTIR Analyses of The Curing Behavior of Epoxy/dicy/solvent Systems on Hermetic Specimens. Journal of Thermal Analysis and Calorimetry 59, 711–719 (2000). https://doi.org/10.1023/A:1010189301221
Issue Date:
DOI: https://doi.org/10.1023/A:1010189301221