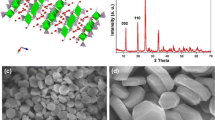
Abstract
The inorganic ion-exchanger α-zirconium phosphate was synthesized by the sol-gel method and its properties relating to the exchange of Cd2+ and the intercalation of CdS particles were studied. The Cd2+-exchange process is a fast process and the material obtained exhibits an increased interlayer distance d with respect to its precursor (9.56 vs. 7.56 Å). The resulting Cd-containing material was exposed to aH2S gas flow to give CdS particles in the exchanger. The zirconium phosphate containing CdS particles still possesses a layered structure, with a pattern almost identical to that of the initial ion-exchanger precursor. Moreover, the material may exchange further Cd2+ and hence lead to a higher CdS particle content. The thermal behavior of this ion-exchangers containing Cd2+ or CdS particles was studied.
Similar content being viewed by others
References
A. Clearfield, Inorganic Ion-Exchange Materials, Ed. A. Clearfield, CRC Press, Boca Raton, FL, 1981, Chap. 1, p. 20 and references therein.
G. Alberti and U. Costantino, Inclusion Compounds, Eds. J. L. Atwood, J. E. D. Davies and D. D. MacNicol, Chap. 5, University Press, New York 1982, p. 136.
A. Clearfield, A. Oskarsson and C. Oskarsson, Ion Exch. Member., 1 (1972) 91; K. A. Kraus and H. O. Phillips, J. Am. Chem. Soc., 78 (1956) 644.
A. Clearfield and J. A. Stynes, J. Inorg. Nucl. Chem., 26 (1964) 117.
G. Alberti and E. Torracca, J. Inorg. Nucl. Chem., 30 (1967) 317.
A. Clearfield and G. Smith, Inorg. Chem., 8 (1969) 431.
J. Livage, H. Benhamza, P. Barboux, A. Bouhaouss and F. A. Josien, J. Mat. Chem., 1 (1991) 681.
A. Clearfield and J. M. Kalnins, J. Inorg. Nucl. Chem., 38 (1976) 849.
S. Allulli, C. Ferragina, A. La Ginestra, M. A. Massucci and N. Tomassini, J. Chem. Soc. Dalton Trans., (1977) 1879.
C. Ferragina, A. La Ginestra, M. A. Massucci, P. Patrono, P. Giannoccaro, F. Nobile and G. Moro, J. Mol. Catal., 53 (1989) 349.
G. Alberti and U. Costantino, Intercalation Chemistry, Eds. M. S. Whittingham and A. J. Jacobson, Academic Press, 1982, p. 147.
W. Chen, Y. Xu, Z. Lin, Z. Wang and L. Lin, Solid State Comm., 105 (1998) 129.
A. G. Cao, L. K. Rabenberg, C. M. Nunn and T. E. Mallouk, Chem. Mat., 3 (1991) 149.
T. Cassageau, G. B. Hix, D. J. Jones, P. Maireles-Torres, M. Rhomari and J. Roziere, J. Mater. Chem., 4 (1994) 189.
C. Ferragina and R. Di Rocco, Mater. Res. Bull., 34 (1999) 1981.
G. Alberti, U. Costantino and E. Torracca, J. Inorg. Chem., 28 (1966) 225.
S. Allulli, C. Ferragina, A. La Ginestra, M. A. Massucci, N. Tomassini and A. A. G. Tomlinson, J. Chem. Soc., (1976) 2115; C. Bettinali, A. La Ginestra and M. Valigi, Atti Accad. Naz. Lincei, Sez. 8, 1962, p. 472.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ferragina, C., Cafarelli, P. & Di Rocco, R. Incorporation of Cadmium Ions and Cadmium Sulfide Particles into Sol-gel Zirconium Phosphate. Synthesis, thermal behavior and X-ray characterization. Journal of Thermal Analysis and Calorimetry 63, 709–721 (2001). https://doi.org/10.1023/A:1010183819410
Issue Date:
DOI: https://doi.org/10.1023/A:1010183819410