Abstract
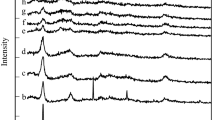
A series of Zn–Al hydrotalcites with Zn/Al molar ratios of 1, 2, 3 and 6 were prepared by co-precipitation method. TG-DTG results showed that the hydrotalcites decompose in two stages, corresponding to the two endothermic peaks around 180 and 220°C. After calcination at 400°C, the samples were converted into Zn–Al mixed oxides with the only XRD pattern of ZnO, except for the sample with the ratio of 6. The Zn–Al mixed oxides possess similar surface acidity revealed by microcalorimetric adsorption of NH3. The basicity of the samples increases with the order: ZnO>6Zn/Al>1Zn/Al>Al2O3.
Similar content being viewed by others
References
F. Cavani, F. Trifiro and F. Vaccari, Catal. Today, 11 (1991) 173.
W. T. Reichle, Chemtech, January (1986) 58.
R. J. Davis and E. G. Derouane, J. Catal., 132 (1991) 269.
E. Narita, P. Kaviratna and T. J. Pinnavaia, Chem. Lett., (1993) 777.
Jianyi Shen, J. M. Kobe, Yi Chen and J. A. Dumesic, Langmuir, 10 (1994) 3902.
G. M. Woltermann, US Patent 4, 454, 244 (1984).
J. M. Lopez, Z. Dejoz and M. I. Vazquez, Appl. Catal., A 132 (1995) 41.
A. Corma and R. M. Martin-Aranda, Appl. Catal., A 105 (1995) 271.
Jianyi Shen, Guang Bin, Mai Tu and Yi Chen, Catal. Today, 30 (1996) 77.
K. Tanabe, M. Misono, T. Ono and H. Hattori, 'Studies in Surface and Catalysis, Vol. 51, Elsevier, Amsterdam 1989.
N. Cardona-Martinez and J. A. Dumesic, Adv. Catal., 38 (1992) 149.
A. Auroux and A. Gervasini, J. Phys. Chem., 94 (1990) 6371.
R. R. Gatte and J. Phillips, J. Catal., 116 (1989) 49.
Mai Tu, Jianyi Shen and Yi Chen, J. Solid Stat. Chem., 128 (1997) 73.
B. E. Handy, S. B. Sharma, B. E. Spiewak and J. A. Dumesic, Meas. Sci. Technol., 4 (1993) 1350.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tu, M., Shen, J. & Chen, Y. Microcalorimetric Studies of Zn—Al Mixed Oxides Obtained From Hydrotalcite-type Precursors. Journal of Thermal Analysis and Calorimetry 58, 441–446 (1999). https://doi.org/10.1023/A:1010171725592
Issue Date:
DOI: https://doi.org/10.1023/A:1010171725592