Abstract
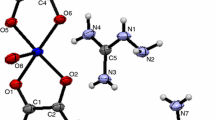
Oxalates of La(III), Ce(III), Pr(III), Nd(III) and Sm(III) with the hydrazinium cation with the general formulae (N2H5)4Ln2(C2O4)57H2O (Ln=La3+, Ce3+, Pr3+) and N2H5Ln(C2O4)2·3.5H2O (Ln=Nd3+, Sm3+) were synthesized. The thermal decompositions of these compounds take place in three stages: thermal dehydration at 65–100°C, exothermic decomposition of the N2H4 at 230–260°C, and oxidation of the oxalate ion.
Similar content being viewed by others
References
O. Genčova and J. Šiftar, Vestn. Slov. Kem. Drus., 38 (1991) 485.
O. Genčova and J. Šiftar, J. Thermal Anal., 44 (1995) 1171.
O. Genčova and J. Šiftar, J. Thermal Anal., 48 (1997) 321.
O. Genčova and J. Šiftar, J. Thermal Anal., 48 (1997) 877.
W. R. Schoellerl and A. R. Powel, The Analysis of Minerals and Ores of the Rare Elements, 3rd Ed., Hafner, New York 1955, p. 105.
W. R. McBride, R. A. Henry and S. Skolnik, Anal. Chem., 23 (1951) 890.
D. Gajapati, S. Govindarajan and K. C. Patil, Thermochim. Acta, 60 (1983) 87.
D. Dellimore and L. Griffiths, J. Thermal Anal., 2 (1970) 229.
W. Balcerowiak, J. Wasilewski and Cz. Latosha, J. Thermal Anal., 18 (1980) 57.
I. A. Kahwa and J. Selbin, J. Thermal Anal., 28 (1983) 359.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Genčova, O., Šiftar, J. Hydrazinium Oxalatometallates of Rare Earths(III). Journal of Thermal Analysis and Calorimetry 57, 591–594 (1999). https://doi.org/10.1023/A:1010148932142
Issue Date:
DOI: https://doi.org/10.1023/A:1010148932142