Abstract
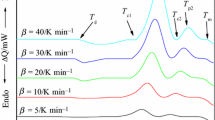
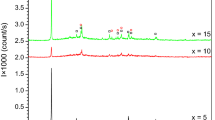
Two glasses of the chalcogenide system Pb20GexSe80-x, with x =17 and 22 at.%, were prepared by the melt quench technique. Differential scanning calorimetry emphasized that the investigated Pb20Ge17Se63 and Pb20Ge22Se58 glasses are crystallized to GeSe2 and PbSe2 as well as GeSe2 and PbSe, respectively as revealed by X-ray diffraction analysis. It was found that the glass transition temperatures of the Pb20Ge22Se58 glass are higher than those of Pb20Ge17Se63 ones. The respective values for the activation energy of glass transition (E t ) for Pb20Ge17Se63 and Pb20Ge22Se58 are found to be 434±20 and 761±77 kJ mol-1, while those for the annealed samples are 928±85 and 508±23 kJ mol-1, respectively. The activation energies of crystallization (E c) before and after annealing were determined using different methods. Applying the modified Johnson-Mehl-Avrami (JMA) equation, it could be found that GeSe2 is crystallized by surface crystallization, while both PbSe2 and PbSe are crystallized by bulk crystallization in three dimensions .
Similar content being viewed by others
References
J. A. Savage, Infrared optical materials and their antireflection coatings, Adam Hiliger, Bristol 1985.
Z. Cimpl and F. Kosek, J. Non-Cryst. Solids, 90 (1987) 577.
A. B. Seddon and M. J. Laine, J. Non-Cryst. Solids, 213 (1997) 168.
N. F. Mott, E. A. Davis and R. Street, Phil. Mag., 32 (1975) 961.
N. Tohge, T. Minami, Y. Yamamoto and M. Tanaka, J. Appl. Phys., 51 (1980) 1048.
N. Tohge, H. Matsuo and T. Minami, J. Non-Cryst. Solids, 95-96 (1987) 809.
K. L. Bhatia, S. K. Malik, N. Kishore and S. P. Singh, Phil. Mag. B, 66 (1992) 587.
B. Vaidhyanathan, S. Murugavel, S. Asokan and K. J. Rao, J. Physical Chemistry B., 101 (1997) 9717.
D. W. Handerson, J. Non-Cryst. Solids, 30 (1979) 301.
M. F. Kotkata and E. A. Mahmoud, Mater. Sci. Engin., 54 (1982) 163.
H. Yinnon and D. R. Uhlmann, J. Non-Cryst. Solids, 54 (1983) 253.
H. E. Kissinger, J. Res. Nat. Bur. Stand, 57 (1956) 217; idem, Anal. Chem., 29 (1957) 1702.
J. A. Augis and J. E. Bennett, J. Thermal Anal., 13 (1978) 283.
K. Matusita, T. Komatsu and R. Yokota, J. Mater. Sci., 19 (1984) 291.
K. Tanaka, Phys. Rev. B, 39 (1989) 1270.
L. Pauling, The Chemical Bond, Cornell University, New York 1976.
R. T. Sanderson, The Chemical Bonds and Bonds Energy, Academic Press, New York 1976.
L. Tichy and H. Ticha, J. Non-Cryst. Solids, 189 (1995) 141.
M. Lasocka, Mat. Sci. Eng., 23 (1976) 173.
J. Colemenero and J. M. Barandiaran, J. Non-Cryst. Solids, 30 (1978) 263.
S. Mahadevan, A. Giridhar and A. K. Singh, J. Non-Cryst. Solids, 88 (1986) 11.
A. Hruby and L. Stourac, Mat. Res. Bull., 6 (1971) 465.
K. Matusita and S. Sakka, Phys. Chem. Glasses, 20 (1979) 81.
J. Malek, Thermochim. Acta, 267 (1995) 61.
R. F. Speyer and S. H. Risbud, Phys. Chem. Glasses, 24 (1983) 26.
M. D. Baro, M. T. Clavaguera-Mora and N. Clavaguera, J. Mater. Sci., 19 (1984) 3005.
N. Afify, J. Non-Cryst. Solids, 126 (1990) 130.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wakkad, M.M. Crystallization Kinetics of Pb20Ge17Se63 and Pb20Ge22Se58 Chalcogenide Glasses. Journal of Thermal Analysis and Calorimetry 63, 533–547 (2000). https://doi.org/10.1023/A:1010146021940
Issue Date:
DOI: https://doi.org/10.1023/A:1010146021940