Abstract
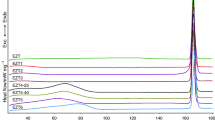
When the DSC analysis of different samples of terpin hydrate is carried out, a non identified small endotherm is observed at about 100°C, just before the melting endotherm. This phenomenon is detected whatever the experimental conditions are.
After some trials, this endotherm was identified as an eutectic formed with terpin hydrate and desolvated terpin (17/83).
The importance of the experimental conditions is preponderant. In an open pan, the desolvation occurs and the melting endotherm of the anhydrous form can be observed at 105°C. In a closed pan, no desolvation is detected and the melting endotherm at 120°C is that of the terpin hydrate.
The eutectic exhibits a good compression ability and a fast dissolution. Its stability is correct. Its use in therapeutic tablets can be envisaged.
The eutectic structure could be, more generally, favourable to compression owing to the isotropic texture of this particular solid state.
Similar content being viewed by others
References
French Pharmacopoeia, 10th Ed.
Merck Index, 11th ed., p. 9105.
'Martindale', the Extra Pharmacopoeia, 27th Ed., The Pharmaceutical Press, London 1977.
J.-C. Guyot, E. Joiris and E. Sergent. 2nd World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology, Paris. May 1998. In Proc. p. 217.
C. Merle, C. Mangin and A.-M. Guyot-Hermann, Bull. Soc. Pharm., Lille, 2–3 (1977) 87.
A. Surlève, D. Leblanc, P. Picot, H. Robert and A.-M. Guyot-Hermann. Bull. Soc. Pharm., Lille, 2 (1980) 143.
JCPDS-ICDD no11 722.
N. School, Natuurw. Tijdschr., 14 (1932) 40.
Rights and permissions
About this article
Cite this article
Di Martino, P., Piva, F., Conflant, P. et al. Thermal Analysis And Powder X-Ray Diffraction Study of Terpin: Evidence of a eutectic ‘hydrate/anhydrous form’. Journal of Thermal Analysis and Calorimetry 57, 95–109 (1999). https://doi.org/10.1023/A:1010120703183
Issue Date:
DOI: https://doi.org/10.1023/A:1010120703183