Abstract
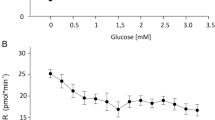
Swimbladder gas gland cells are polar epithelial cells which release acidic metabolites through the membranes of an extensive basolateral labyrinth, and secret surfactant via exocytosis at apical membranes. We have developed a method to establish primary cell cultures of gas gland cells in order to establish a model system for physiological analysis of gas gland cell function in vitro. Isolated gas gland cells attach to collagen S coated surfaces. Cells cultured in collagen S coated petri dishes were flat and showed no histological polarity. Cells cultured on Anodisc membranes in a superfusion system, in which the apical and basal side of the cells was supplied with a saline solution and with glucose containing DMEM cell culture medium, respectively, showed a clear polarity similar to the in vivo situation. Measurement of lactate release at the apical side and at the basal side revealed that these cells were functionally polar and secreted at least 70% of the lactate at their basal membranes. Gas gland cells could also be cultured in an air/liquid system, in which the apical membrane was exposed to humidified air. Cells cultured under these conditions released lactate only on the basal side and histologically were similar to cells cultured in the superfusion system.
Similar content being viewed by others
References
Bergmeyer H-U (ed) (1974). Methoden der enzymatischen Analyse. Weinheim: Verlag Chemie, 2353 pp.
Daniels CB, Skinner CH (1994). The composition and function of surface-active lipids in the goldfish swim bladder. Physiol Zool 67: 1230-1256.
Dorn E (1961). Ñber den Feinbau der Schwimmblase von Anguilla vulgaris L. Licht-und Elektronenmikroskopische Untersuchungen. Z Zellforsch 55: 849-912.
Fänge R (1983). Gas exchange in fish swim bladder. Rev Physiol Biochem Pharmacol 97: 111-158.
Kuhn W, Ramel A, Kuhn HJ, Marti E (1963). The filling mechanism of the swimbladder. Generation of high gas pressures through hairpin countercurrent multiplication. Experientia 19: 497-511.
Mair N, Haller T, Dietl P (1999). Exocytosis in alveolar type II cells revealed by cell capacitance and fluorescence measurements. Am J Physiol 276: L376-L382.
Mason RJ, Shannon JM (1997). Alveolar type II cells. In: Crystal RG, West JB et al. (eds), The Lung: Scientific Foundations, pp 543-555. Philadelphia: Lippincott-Raven Publishers.
Pelster B (1994). Adrenergic control of swimbladder perfusion in the European eel Anguilla anguilla. J exp Biol 189: 237-250.
Pelster B (1995). Metabolism of the swimbladder tissue. Biochem Molec Biol Fishes 4: 101-118.
Pelster B (1997). Buoyancy at depth. In: Randall D, Farrell AP (eds), Deep-Sea Fish, pp 195-237. San Diego: Academic Press.
Pelster B, Hicks J, Driedzic WR (1994). Contribution of the pentose phosphate shunt to the formation of CO2 in swimbladder tissue of the eel. J exp Biol 197: 119-128.
Pelster B, Pott L (1996). Control of acid secretion in cultured gas gland cells of the European eel Anguilla anguilla. Am J Physiol 270: R578-R584.
Pelster B, Randall DJ (1998). The physiology of the Root effect. In: Perry SF, Tufts BL (eds), Fish Respiration, pp 113-139. San Diego: Academic Press.
Pelster B, Scheid P (1992). The influence of gas gland metabolism and blood flow on gas deposition into the swimbladder of the European eel Anguilla anguilla. J exp Biol 173: 205-216.
Pelster B, Weber RE (1991). The physiology of the Root effect. Adv Comp Environm Physiol 8: 51-77.
Prem C, Salvenmoser W, Pelster B (1999). Surfactant production in swimbladder gas gland cells: in situ and in culture. Zoology 102: 71.
Walsh PJ, Milligan CL (1993). Roles of buffering capacity and pentose phosphate pathway activity in the gas gland of the gulf toadfish Opsanus beta. J exp Biol 176: 311-316.
Würtz J, Salvenmoser W, Pelster B (1999). Localization of carbonic anhydrase in swimbladder tissue of European eel (Anguilla anguilla) and perch (Perca fluviatilis). Acta Physiol Scand 165: 219-224.
Rights and permissions
About this article
Cite this article
Prem, C., Pelster, B. Swimbladder gas gland cells of the European eel cultured in a superfusion system. Methods Cell Sci 22, 125–132 (2000). https://doi.org/10.1023/A:1009826424171
Issue Date:
DOI: https://doi.org/10.1023/A:1009826424171