Abstract
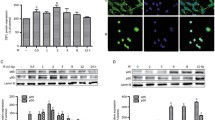
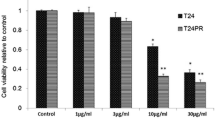
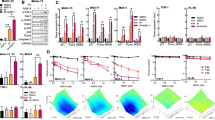
Fractionated γ-irradiation (15 × 2 Gy in 3 weeks) induces a cellular resistance in HeLa cells against cisplatin exposure but not against irradiation. The mechanisms underlying this cellular resistance are associated with major changes in the TNFR1-dependent transduction pathway. The resistant HeLa/B cells exhibit increased levels of NF-κB with temporally independent regulation of the subunits NF-κB50 and NF-κB65. Blocking IκB degradation by the proteasome inhibitor PSI, which abolishes the release of the active NF-κB protein, induces cell death much more effectively in the parental than in the resistant HeLa/B cells. The translocation of NF-κB does not seem to be affected in a similar manner since masking of the translocation sequence by NF-κB SN50 enhances cisplatin toxicity to the same degree in both cell lines and overcomes drug resistance. Changes in upstream signaling are suggested by increased sensitivity of the parental HeLa cells to cisplatin in the presence of neutralizing anti-TNFR1. In HeLa/B cells, reduced expression of the 50 kDa silencer of death domain, SODD, is accompanied by constitutive overexpression of a 40–42 kDa SODD-like protein. A possible involvement of SODD in cisplatin resistance is discussed, which may shift the balance between life and death in the TNF receptor pathway to increased NF-κB activation.
Similar content being viewed by others
References
Eichholtz-Wirth H, Marx K. Clonal variability of radiationinduced cisplatin resistant HeLa cells. Anticancer Res 1998; 18: 2989–2992.
May MJ, Gosh S. Signal transduction through NF-· B. Rev Immunology Today 1998; 19: 80–88.
Chen ZJ, Parent L, Maniatis T. Site-specific phosphorylation of IkBa by a novel ubiquitination-dependent protein kinase activity. Cell 1996; 84: 853–862.
Palaombella VJ, Rando OJ, Goldberg AL, Maniatis T. The ubiquitin-proteasome pathway is required for processing the NF-·B 1 precursor protein and the activation of NF-·B. Cell 1994; 78: 773–785.
May MJ, Gosh S. Rel/NF-kB and IkB proteins: An overview. Sem Cancer Biol 1997; 8: 63–73.
Naumann M, Scheidereit C. Activation of NF-·B in vivo is regulated by multiple phosphorylations. EMBO J 1994; 4597–4607.
Mercurio F, Zhu H, Murray BW, et al. IKK-1 and IKK-2: Cytokine-activated IkB kinases essential for NF-·B activation. Science 1997; 278: 860–866.
Hsu H, Shu HB, Pan MG, Goeddel DV. TRADD-Traf2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal pathways. Cell 1996; 84: 299–308.
Hsu H, Huang J, Shu HB, Baichwal V, Goeddel DV. TNFdependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity 1996; 4: 387–396.
Kelliher MA, Grimm S, Ishida Y, Kuo F, Stanger BZ, Leder P. The death domain kinase RIP mediates the TNF-induced NF-·B signal. Immunity 1998; 8: 297–303.
Jiang Y, Woronicz JD, Liu W, Goeddel DV. Prevention of constitutive TNF receptor 1 signaling by silencer of death domain. Science 1999; 283: 543–546.
Lin Y, Devin A, Rodriguez Y, Liu Z. Cleavage of the death domain kinase RIP by caspase-8 prompts TNF-induced apoptosis. Genes Dev 1999; 13: 2514–2526.
Eichholtz-Wirth H, Stoetzer O, Marx K. Reduced expression of the ICE-related protease CPP32 is associated with radiationinduced cisplatin resistance in HeLa cells. Br J Cancer 1997; 76: 1322–1327.
Lee DH, Goldberg AL. Proteasome inhibitors: Valuable tools for cell biologists. Tr Cell Biol 1998; 8: 397–403.
Tschopp J, Martinon F, Hofmann K. Apoptosis: Silencing of death receptors. Current Biology 1999; 9: R381–R384.
Fisher DE. Apoptosis in cancer therapy: Crossing the threshold. Cell 1994; 78: 539–542.
Lin KI, DiDonato JA, Hoffmann A, Hardwick JM, Ratan RR. Suppression of the steady-state, but not stimulus-induced NF-·B activity inhibits alphavirus-induced apoptosis. J Cell Biol1998; 141: 1479–1487.
Grimm S, Bauer MK, Baeuerle PA, Schulze-Osthoff K. Bcl-2 down-regulates the activity of transcription factor NF-·B induced upon apoptosis. J Cell Biol 1996; 134: 13–23.
Lin Y, Yao S, Veach RA, Torgerson TR, Hawiger J. Inhibition of nuclear translocation of transcription factor NF-·B by a synthetic peptide containing a cell membrane-permeable motif and nuclear localization sequence. J Biol Chem 1995; 270: 14255–14258.
Takayama S, Sata T, Krajewski S, et al. Cloning and functional analysis of BAG-1: A novel Bcl-2-binding protein with anticell death activity. Cell 1995; 80: 279–284.
Takayama S, Xie Z, Reed JC. An evolutionary conserved family of Hsp70/Hsc70 molecular chaperone regulators. J Biol Chem 1999; 274: 781–786.
Rothwarf DM, Zandi E, Natoli G, arin M. IKK-° is an essential regulatory subunit of the I·B kinase complex. Nature 1998; 395: 297–300.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eichholtz-Wirth, H., Sagan, D. IκB/NF-κB mediated cisplatin resistance in HeLa cells after low-dose γ-irradiation is associated with altered SODD expression. Apoptosis 5, 255–263 (2000). https://doi.org/10.1023/A:1009656513307
Issue Date:
DOI: https://doi.org/10.1023/A:1009656513307