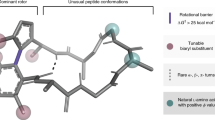
Abstract
The crystal structures of two molecular propellers are described. Enol phosphate 1 is triclinic P 1¯ with a = 10.1110(4), b = 12.0647(6), c = 14.2112(7) Å; α = 96.421(3), β = 98.633(3), γ = 108.594(2)° and Z = 2. Enol phosphinate 2 is monoclinic P21/n with a = 13.527(3), b = 11.980(2), c = 17.326(3) Å, β = 98.616(9),° and Z = 4. Data from dynamic NMR investigations are in agreement with the size of the aryl torsion angles in the two enol propellers.
Similar content being viewed by others
References
Mislow, K. Acc. Chem. Res. 1976, 9, 26.
Luef, W.; Keese, R. Top. Stereochem. 1991, 20, 231.
Biali, S.E.; Rappoport, Z. J. Am. Chem. Soc. 1984, 106, 477.
Rappoport, Z.; Biali, S.E. Acc. Chem. Res. 1988, 21, 442. (b) Kaftory, M.; Nugiel, D.A.; Biali, S.E.; Rappoport, Z. J. Am. Chem. Soc. 1989, 111, 8181. (c) Schottland, E.; Frey, J.; Rappoport, Z. J. Org. Chem. 1994, 59, 1663 and references cited therein.
Schmittel, M.; Steffen, J.-P.; Burghart, A. J. Chem. Soc. Chem. Commun. 1996, 2349.
Schmittel, M.; Keller, M.; Burghart, A. J. Chem. Soc. Perkin Trans. 2 1995, 2327.
Schmittel, M.; Heinze, J.; Trenkle, H. J. Org. Chem. 1995, 60, 2726.
Schmittel, M.; Trenkle, H. Chem. Lett. 1997, 299.
Schmittel, M.; Söllner, R. Chem. Ber./Recueil 1997, 130, 771.
Röck, M.; Schmittel, M. J. Prakt. Chem. 1994, 336, 325.
Nadler, E.B.; Röck, M.; Schmittel, M.; Rappoport, Z. J. Phys. Org. Chem. 1993, 6, 233.
Schmittel, M.; Keller, M.; Burghart, A.; Rappoport, Z.; Langels, A. unpublished results.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burghart, A., Schmittel, M. & Keller, M. Structures of two sterically crowded molecular propeller systems. Journal of Chemical Crystallography 29, 421–427 (1999). https://doi.org/10.1023/A:1009563026959
Issue Date:
DOI: https://doi.org/10.1023/A:1009563026959