Abstract
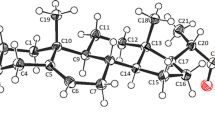
Two epimers of 20-aminopregn-5-en-3β-ol differing in configuration at C(20) have been synthesized and their structures have been determined by single crystal X-ray diffraction methods. The (20R)-epimer (1) crystallized in the orthorhombic system, space group P212121, with a = 9.0432(5) Å, b = 13.3446(7) Å, c = 15.1086(8) Å, and Z = 4. The (20S)-epimer hydrochloride hydrate (2 hydrate) crystallized in the monoclinic system, space group C2, with a = 31.0501(3) Å, b = 11.8221(1) Å, c = 11.9703(2) Å, β = 98.5140(10)°, and Z = 8. The absolute configurations at the C(20) chiral centers for the title compounds were unequivocally established for the first time. The asymmetric unit cell of 2 hydrate contained two steroid molecules, A and B, two hydrochloride molecules, and two molecules of water. The crystal packing for both diastereomers was strongly influenced by the presence of intermolecular hydrogen bonding. The molecules of 1 were linked in a head-to-tail fashion via hydrogen bonds between the hydroxyl group of the ring A and the amino group of the side chain of the ring D. The two molecules A and B of 2 hydrate were held together in head-to-head and head-to-tail fashions by a three-dimensional network of hydrogen bonds involving chlorine ions, disordered water molecules, and hydroxyl–amino groups interactions.
Similar content being viewed by others
References
Sheets, J.J.; Vickery, L.E. J. Biol. Chem. 1983, 258, 11446.
Kellis, J.T., Jr.; Sheets, J.J.; Vickery, L.E. J. Steroid Biochem. 1984, 20, 671.
Li, J.; Li, Y.; Son, C.; Brodie. A.M.H. J. Med. Chem. 1996, 39, 4335.
Njar, V.C.O.; Hector, M.; Hartmann, R.W. Bioorg. Med. Chem. 1996, 4, 1447.
Ling, Y.; Li, J.; Kato, K.; Liu, Y.; Wang, X.; Klus, G.T.; Marat, K.; Nnane, I.P.; Brodie, A.M.H. Bioorg. Med. Chem. 1998, 6, 1683.
Chavis, C.; de Gourcy, C.; Borgna, J.-L.; Imbach, J.-L. Steroids 1982, 39, 129.
Biesemans, M.; Van de Woude, G.; Van Hove, L. Bull. Soc. Chim. Belg. 1985, 94, 59.
Lucas, R.A.; Dickel, D.F.; Dziemian, R.L.; Ceglowski, M.J.; Hensle, B.L.; MacPhillamy, H.B. J. Am. Chem. Soc. 1960, 82, 5688.
Van de Woude, G.; Van Hove, L. Bull. Soc. Chim. Belg. 1967, 76, 566.
Lu, M.C.; Afiatpour, P.; Sullivan, C.B.; Counsell, R.E. J. Med. Chem. 1972, 15, 1284.
Goutarel, R.; Mahler, H.R.; Green, G.; Khuong-Huu, Q.; Cavé, A.; Conreur, C.; Jarreau, F.-X.; Hannart, J. Bull. Soc. Chim. Fr. 1967, 12, 4575.
Szendi, Z.; Dombi, G.; Vincze, I. Monatsh. Chem. 1996, 127, 1189.
Sheldrick, G.M. SHELXTL, Version 5.10; Bruker AXS Inc.: Madison, WI, USA, 1998.
SMART, Version 5.054; Bruker AXS Inc.: Madison, WI, USA, 1998.
SAINT, Version 6.01; Bruker AXS Inc.: Madison, WI, USA, 1999.
Spek, A.L. PLATON; Utrecht University, The Netherlands, Windows Version by Farrugia, L.J., University of Glasgow, United Kingdom, 1999.
Duax, W.L.; Norton, D.A. Atlas of Steroid Structure; Plenum: New York, 1975; Vol. 1.
Griffin, J.F.; Duax, W.L.; Weeks, C.M. Atlas of Steroid Structure; Plenum: New York, 1984; Vol. 2.
Thomas, S.A. J. Crystallogr. Spectrosc. Res. 1982, 12, 171.
Bucourt, R. Topics in Stereochemistry; Eliel, E.L.; Allinger, N.L., Eds., Interscience: New York, 1974; Vol. 8, p. 159.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gruszecka-Kowalik, E., VanDerveer, D.G. & Zalkow, L.H. Crystal and molecular structures of (20R)-20-aminopregn-5-en-3β-ol and (20S)-20-aminopregn-5-en-3β-ol hydrochloride. Journal of Chemical Crystallography 30, 291–299 (2000). https://doi.org/10.1023/A:1009553022723
Issue Date:
DOI: https://doi.org/10.1023/A:1009553022723