Abstract
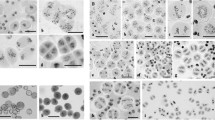
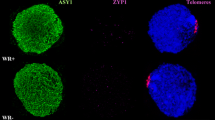
The neocentric activity of a constriction located on the long arm of rye 5R chromosome (5RL) was analysed. It is not observed in normal rye but it is unusually stretched in bivalents involving 5RL telosomes in wheat–ditelosomic 5RL addition lines. In 20% of metaphase I cells, the 5RL bivalent presents the centromeres oriented to one pole and the constrictions oriented towards the opposite pole with a strong tension. In 5% of the cells, the constriction was able to orient the bivalent to the poles without tension in the centromeres. Sister chromatid cohesion, which is one of the distinct features of centromeric function, is persistent at the constriction in delayed 5RL chromosomes at anaphase I. Neither the elongation of the constriction nor the neocentric activity was observed at second meiotic division or mitosis. FISH studies showed that the 5RL constriction lacked detectable quantities of two repetitive DNA sequences, CCS1 and the 180-bp knob repeat, present at cereal centromeres and neocentromeres, respectively. We propose that, under special conditions, such as the wheat background, the normally non-centromeric DNA present at this region of 5RL acquires a specific chromatin structure, differentiated as an elongated constriction, which is able to function as a centromere.
Similar content being viewed by others
REFERENCES
Ananiev EV, Phillips RL, Rines HW (1998) Chromosome-specific molecular organization of maize (Zea mays L.) centromeric regions. Genetics 95: 13073-13078.
Aragón-Alcaide L, Miller T, Schwarzacher T, Reader S, Moore G (1996) A cereal centromeric sequence. Chromosoma 105: 261-268.
Bajer A (1968) Behaviour and fine structure of spindle fibers during mitosis in endosperm. Chromosoma 25: 249-281.
Barry AE, Howman EV, Cancilla MR, Saffery R, Choo KHA (1999) Sequence analysis of an 80 kb human neocentromere. Hum Mol Genet 8: 217-227.
Brown W, Tyler-Smith C (1995) Centromere activation. Trends Genet 11: 337-339.
Cancilla MR, Tainton KM, Barry AE et al. (1998) Direct cloning of human 10q25 neocentromere DNA using transformation associated recombination (TAR) in yeast. Genomics 47: 399-404.
Choo KHA (1997) Centromere DNA dynamics: latent centromeres and neocentromere formation. Am J Hum Genet 61: 1225-1233.
Choo KHA (1998) Turning on the centromere. Nature Genet 18: 3-4.
Dawe RK, Reed LM, Yu H-G, Muszynski MG, Hiatt EN (1999) A maize homolog of mammalian CENPC is a constitutive component of the inner kinetochore. Plant Cell 11: 1227-1238.
Heyward MD (1962) Genetic control of neocentric activity in rye. Heredity 17: 439-441.
Jones G (1969) Further correlations between chiasmata and U-type exchanges in rye meiosis. Chromosoma 26: 105-118.
Karpen GH, Allshire RC (1997) The case for epigenetic effect on centromere identity and function. Trends Genet 13: 489-496.
Katterman G (1939) Ein neuer karyotyp bei roggen. Chromosoma 1: 284-299.
Kavander T, Vinikka Y (1987) Neocentric activity in open-pollinated cultivars of rye. Hereditas 107: 229-233.
Mikhailova EI, Naranjo T, Shepherd K, Wennekes van Eden J, Heyting C, de Jong JH (1998) The effect of the wheat Ph1 locus on chromatin organisation and meiotic chromosome pairing analysed by genome painting. Chromosoma 107: 338-350.
Moore G, Aragón-Alcaide L, Roberts M, Reader S, Miller T, Foote T (1997a) Are rice chromosomes components of a holocentric chromosome ancestor? Plant Mol Biol 35: 17-23.
Moore G, Roberts M, Aragón-Alcaide L, Foote T (1997b) Centromeric sites and cereal chromosome evolution. Chromosoma 105: 321-323.
Moore DP, Page AW, Tang TT-L, Kerrebrock AW, Orr-Weaver TL (1998) The cohesion protein MEI-S332 localizes to condensed meiotic and mitotic centromeres until sister chromatids separate. J Cell Biol 140: 1003-1012.
Peacock WJ, Dennis ES, Rhoades MM, Pryor AJ (1981) Highly repeated DNA sequence limited to knob heterochromatin in maize. Proc Natl Acad Sci USA 78: 4490-4494.
Prakken R, Müntzing A (1942) A meiotic peculiarity in rye, simulating a terminal centromere. Hereditas 28: 441-482.
Presting GG, Malysheva L, Fuchs J, Schubert I (1998) A Ty3/gypsy retrotransposon-like sequence localizes to the centromeric regions of cereal chromosomes. Plant J 16: 721-728.
Rhoades MM, Vikomerson H (1942) On the anaphase movement of chromosomes. Proc Natl Acad Sci USA 28: 433-436.
Schlegel R (1987) Neocentric activity in chromosome 5R of rye revealed by haploidy. Hereditas 107: 1-6.
Sekelsky JJ, Hawley RS (1995) The bond between sisters. Cell 83: 157-160.
Sybenga J (1981) Specialization in the behaviour of chromosomes on the meiotic spindle. Genetica 57: 143-151.
Vinikka Y (1985) Identification of the chromosomes showing neocentric activity in rye. Theor Appl Genet 70: 66-71.
Vinikka Y, Kavander T (1986) C-band polymorphism in the inbred lines showing neocentric activity in rye. Hereditas 104: 203-207.
Warburton PE, Cooke HJ (1997) Hamster chromosomes containing amplified human a satellite DNA show delayed sister chromatid separation in the absence of de nova kinetochore formation. Chromosoma 106: 149-159.
Willard HF (1990) Centromeres of mammalian chromosomes. Trends Genet 6: 410-416.
Williams BC, Murphy TD, Goldberg ML, Karpen GH (1998) Neocentromere activity of structurally acentric minichromosomes in Drosophila. Nature Genet 18: 30-37.
Yu H-G, Hiatt EN, Chan A, Sweeney M, Dawe RK (1997) Neocentromere-mediated chromosome movement in maize. J Cell Biol 139: 831-840.
Zhong X, Fransz PF, Wennekes van Eden J, Zabel P, van Kammen A, de Jong JH (1996) High-resolution mapping on pachytene chromosomes and extended DNA fibres by fluorescent in situ hybridisation. Plant Mol Biol Rep 14: 232-242.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Manzanero, S., Puertas, M.J., Jiménez, G. et al. Neocentric activity of rye 5RL chromosome in wheat. Chromosome Res 8, 543–554 (2000). https://doi.org/10.1023/A:1009275807397
Issue Date:
DOI: https://doi.org/10.1023/A:1009275807397