Abstract
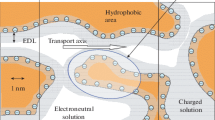
A physicochemical model for the transfer of amino acids through the interface between a cation-exchange membrane and an amino acid aqueous solution is proposed. The model allows for a heterogeneous reaction of protonation of the amino acid zwitterion. The model calculations are compared with experimentally determined migration transport numbers for glycine cations in an electromembrane system with an MK-40 membrane. The comparison shows that, owing to the protonation reaction, the glycine flux can increase by ca. 30%.
Similar content being viewed by others
REFERENCES
Khoshkbarchi, N. and Vera, J., Ind. Eng. Chem. Res., 1996, vol. 35, p. 2735.
Mulder, M., Basic Principles of Membrane Technology, Dordrecht: Kluwer, 1996. Translated under the title Vvedenie v membrannuyu tekhnologiyu, Moscow: Mir, 1999.
Sikdar, S., J. Membr. Sci., 1985, vol. 24, p. 59.
Metayer, M., Langevin, D., Mahi, B., and Pinoche, M., J. Membr. Sci., 1991, vol. 61, p. 191.
Rogers, J. and Long, R., J. Membr. Sci., 1997, vol. 134, p. 1.
Timashev, S.F., Fizikokhimiya membrannykh protsessov (Physical Chemistry of Membrane Processes), Moscow: Khimiya, 1988.
Timashev, S.F., Zh. Fiz. Khim., 1982, vol. 56, p. 1739.
Aristov, I.V., Bobreshova, O.V., and Eliseev, S.Ya., Teor. Prakt. Sorbtsionnykh Protsessov, 1999, no. 24, p. 26.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aristov, I.V., Bobreshova, O.V., Kulintsov, P.I. et al. Transfer of Amino Acids through a Membrane/Solution Interface in the Presence of Heterogeneous Chemical Protonation Reaction. Russian Journal of Electrochemistry 37, 218–221 (2001). https://doi.org/10.1023/A:1009000529087
Issue Date:
DOI: https://doi.org/10.1023/A:1009000529087