Abstract
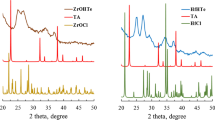
Sulfated zirconia catalysts were prepared by sol-gel method using CH3COOH as in situ water source to control hydrolysis of alkoxide, and following two sulfation procedures. The samples were characterized by N2 adsorption, XRD, chemical analysis, and the activity for isomerization of n-hexane was assessed. It was found that sulfation procedure and the amount of acetic acid added exert a great influence on the catalysts properties. Mixture of sulfuric acid with zirconium propoxide before addition of acetic acid allows the retention of a larger amount of sulfur after calcination and enhances catalytic performances of sulfated zirconia. The use of CH3COOH reduces the rate of hydrolysis, and improves considerably acidic and catalytic properties.
Similar content being viewed by others
References
Bo-Qing Xu and Wolfgang M.H. Sachtler, Journal of Catalysis 167, 224 (1997).
C. Morterra, G. Cerrato, F. Pinna, and M. Signoretto, Journal of Catalysis 157, 109 (1995).
David A. Ward and Edmond I. Ko, Journal of Catalysis 150, 18 (1994).
S. Ardizzone, C. L. Bianchi, and M. Signoretto, Applied Surface Science 136, 213 (1998).
Robert A. Keogh, Ram Srinivasan, and Burton H. Davis, Journal of Catalysis 151, 292 (1995).
Steven X. Song and Ronald A. Kydd, J. Chem. Soc. Faraday Trans. 94(9), 1333 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hamouda, L.B., Ghorbel, A. Control Preparation of Sulfated Zirconia by Sol-Gel Process: Impact on Catalytic Performances During n-Hexane Isomerization. Journal of Sol-Gel Science and Technology 19, 413–416 (2000). https://doi.org/10.1023/A:1008787111207
Issue Date:
DOI: https://doi.org/10.1023/A:1008787111207