Abstract
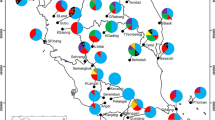
Genetic variation and its distribution within and among 23 populations of Triticum urartu collected from Syria, Lebanon, Turkey, Armenia, and Iran was estimated using isozyme markers at eight polymorphic loci. The number of alleles per locus (A= 1.21), percentage polymorphic loci (P= 20.1%), and mean gene diversity (He= 0.024) were relatively low. In a population from Lebanon, a high number of alleles per locus (A= 2.13) and percentage polymorphic loci (P= 87.5%) was found. On average, genetic variation among populations (GST= 0.407) was smaller than within-population variation (0.593). However, different patterns of genetic structure were found among various geographic regions. Interpopulation variation was highest for the Iranian populations (0.89) followed by the Turkish populations (0.66). A reverse pattern was observed for the Syrian (0.11) and for the Lebanese (0.13) populations. The Armenian populations exhibited similar interpopulation and within-population variation. Principal component and cluster analyses resulted in distinct grouping of the geographically proximal populations, with the exception of the two Iranian populations. The Turkish populations were different from the neighboring Armenian populations compared to other countries. The populations from southern Syria and those from Lebanon also exhibited a high degree of genetic diversity. The two most heterozygous loci, Mdh-2 and Pgi-2, separated the populations along the first and second principal components, respectively. Most of the rare alleles were scattered sporadically throughout the geographic regions. Rare alleles with high frequencies were found in the Turkish and Armenian populations. These results indicated that different geographic regions require specific sampling procedures in order to capture the range of genetic variation observed in T. urartu populations.
Similar content being viewed by others
References
Brown, A.D.H., 1978. Isozymes, plant population genetic structure and genetic conservation. Theor. Appl. Genet. 52: 145–157.
Brown, A.H.D. & J.J. Burdon, 1987. Mating systems and colonizing success in plants. In: Gray, A.J., M.J. Crawley & P.J. Edwards (Eds.), Colonization, Succession and Stability, pp. 115–131. Blackwell Scientific Publication, U.K.
Castagna, R., S. Gnocchi, M. Perenzin & M. Heun, 1997. Genetic variability of the wild diploid wheat Triticum urartu revealed by RFLP and RAPD markers. Theor. Appl. Genet. 94: 424–430.
Chapman, V., T.E. Miller & R. Riley, 1976. Equivalence of the A genome of bread wheat and that of T. urartu. Genet. Res. 27: 69–76.
Cox, T.S., 1991. The contribution of introduced germplasm to the development of U.S. wheat cultivars. In: Shands, H.L. & L.E. Wiesner (Eds.), Use of Plant Introductions in Cultivar Development, pp. 25-47, Part 1, CSSA Special Publication No. 17, Madison, WI, USA.
Dhaliwal, H.S. & B.L. Johnson, 1976. Anther morphology and the origin of the tetraploid wheats. Am. J. Bot. 63: 363–368.
Dhaliwal, H.S., J.S. Sidhu & J.L. Minocha, 1993. Genetic diversity in diploid and hexaploid wheats as revealed by RAPD markers. Crop. Improv. 20: 17–20.
Dudnikov, A.J.U., 1998. Allozyme variation in Transcaucasian populations of Aegilops squarrosa. Heredity 80: 248–258.
D'Ovidio, R., O.A. Tanzarella, A. Cenci, E. Iacono & E. Porceddu, 1994. RFLP analysis in wheat. Isolation and chromosomal assignment of digoxigenin-labelled clones. J. Genet. Breed. 48: 73–80.
Dvorák, J., 1976. The relationship between the genome of Triticum urartu and the A and B genomes of Triticum aestivum. Can. J. Genet. Cytol. 18: 371–377.
Dvorák, J., P.E. McGuire & B. Cassidy, 1988. Apparent sources of the A genomes of wheats inferred from polymorphism in abundance and restriction fragment length of repeated nucleotide sequences. Genome 30: 680–689.
Dvorák, J., P. Di Terlizzi, H.B. Zhang & P. Resta, 1993. The evolution of polyploid wheats: identification of the A genome donor species. Genome 36: 21–31.
Ellstrand, N.C., 1984. Multiple paternity within the fruits of the wild radish, Raphanus sativus. Am. Nat. 123: 819–828.
Firat, A.E. & A. Tan, 1998. Ecogeography and distribution of wild cereals in Turkey. In: Zencirci, N., K. Kaya, Y. Anikster & W.T. Adams (Eds.), Proc. Int. Symp. In Situ Conservation of Plant Genetic Diversity, pp. 81–85. Central Res. Inst. For Field Crops, Ankara, Turkey.
Gale, M.D. & P.J. Sharp, 1988. Genetic markers in wheat-developments and prospects, pp. 469-475. Proc. 7th. Int. Wheat Genet. Symp., Cambridge, U.K.
Garvin, D.F., M.L. Roose & J.G. Waines, 1989. Isozyme genetics and linkage in tepary bean, Phaseolus acutifolis A. Gray. J. Hered. 80: 373–376.
Hamrick, J.L., Y.B. Linhart & J.B. Mitton, 1979. Relationships between life history characteristics and electrophoreticallydetectable genetic variation in plants. Annu. Rev. Ecol. Syst. 10: 173–200.
Harlan, J.R, 1976. Genetic resources in wild relatives of crops. Crop Sci. 16: 329–333.
Jaaska, V., 1993. Isoenzymes in the evaluation of germplasm diversity in wild diploid relatives of cultivated wheat. In: Damania, A.B. (Ed.), Biodiversity and Wheat Improvement, pp. 247–257. John Wiley & Sons, New York, NY.
Jakubizner, M.M., 1959. New wheat species. In: Jenkins, BC (Ed.), pp. 207-217. Proc. 1st Int. Wheat Genet. Symp., Winnipeg, Canada.
Johnson, B.L., 1975. Identification of the apparent B-genome donor of wheat. Can. J. Genet. Cytol. 17: 21–39.
Johnson, B.L. & H.S. Dhaliwal, 1978. Triticum urartu and genome evolution in tetraploid wheats. Am. J. Bot. 65: 907–918.
Kimber, G. & E.R. Sears, 1987. Evolution in the genus Triticum and the origin of cultivated wheat. In: Heyne, E.G. (Ed.), Wheat and Wheat Improvement, pp. 154–163. American Society of Agronomy, Madison, WI, USA.
Le Corre, V. & M. Bernard, 1995. Assessment of the type and degree of restriction fragment length polymorphism (RFLP) in diploid species of the genus Triticum. Theor. Appl. Genet. 90: 1063–1067.
Lewis, P.O., 1992. GeneStat-PC 3.3. Department of Statistics, Raleigh, North Carolina University, NC, USA.
Marshall, D.R. & R.W. Allard, 1970. Maintenance of isozyme polymorphism in natural populations of Avena barbata. Genetics 66: 393–399.
Nei, M., 1973. Analysis of gene diversity in sub-divided populations. Proc. Natl. Acad. Sci. USA 70: 3321–3323.
Nei, M., 1978. Estimation of average heterozygosity and genetic distance from small number of individuals. Genetics 89: 583–590.
Poulik, M.D., 1957. Starch gel electrophoresis in discontinuous system of buffers. Nature 180: 1477–1479.
Rafi, M.M., B. Ehdaie & J.G. Waines, 1992. Quality traits, carbon isotope discrimination and yield components in wild wheats. Ann. Bot. 69: 467–474.
Rohlf, F.J., 1993. Numerical taxonomy and multivariate analysis system, version 1.80. 100 North Country Road, Setauket, NY.
Skovmand, B. & S. Rajaram, 1990. Utilization of genetic resources in the improvement of hexaploid wheat. In: Srivastava, J.P. & A.B. Damania (Eds.), Wheat genetic resources: meeting diverse needs, pp. 259–268. John Wiley & Sons, New York, NY.
Slatkin, M. & N.H. Barton, 1989. A comparison of three indirect methods for estimating average levels of gene flow. Evolution 43: 1349–1368.
Smith-Huerta, N.L., A.J. Huerta, D. Barnhart & J.G. Waines, 1989. Genetic diversity in wild diploid wheats Triticum monococcum var. boeoticum and T. urartu (Poaceae). Theor. Appl. Genet. 78: 260–264.
Soltis, D.E., C.H. Haufler, D.C. Darrow & G.J. Gastony, 1983. Starch gel electrophoresis of fern: a compilation of grinding buffers, gel and electrode buffers, and staining schedules. Am. Fern. J. 73: 9–27.
Takumi, S., S. Nasuda, Y.G. Liu & K. Tsunewaki, 1993. Wheat phylogeny determined by RFLP analysis of nuclear DNA. 1. Einkorn wheat. Jpn. J. Genet. 68: 73–79.
Vierling, R.A. & H.T. Nguyen, 1992. Use of RAPD markers to determine the genetic diversity of diploid wheat genotypes. Theor. Appl. Genet. 84: 835–838.
Waines, J.G. & P.I. Payne, 1987. Electrophoretic analysis of the high-molecular-weight glutenin subunits of Triticum monococcum, T. urartu and the A genome of bread wheat (T. aestivum). Theor. Appl. Genet. 74: 71–76.
Waines, J.G., B. Ehdaie & D. Barnhart, 1987. Variability in Triticum and Aegilops species for seed characteristics. Genome 29: 41–46.
Wendel, J.F. & N.F. Weeden, 1989. Visualization and interpretation of plant isozymes. In: Soltis, D.E. & P.S. Soltis (Eds.), Isozymes in Plant Biology, pp. 5–45. Dioscorides Press, OR, USA.
Yaghoobi-Saray, J., 1979. An electrophoretic analysis of genetic variation within and between populations of five species in Triticum-Aegilops complex. Ph.D. Thesis, University of California, Davis, CA, USA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moghaddam, M., Ehdaie, B. & Waines, J.G. Genetic diversity in populations of wild diploid wheat Triticum urartu Tum. ex. Gandil. revealed by isozyme markers. Genetic Resources and Crop Evolution 47, 323–334 (2000). https://doi.org/10.1023/A:1008782532505
Issue Date:
DOI: https://doi.org/10.1023/A:1008782532505