Abstract
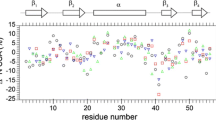
Backbone dynamics of uniformly 15N-labeled free barnase and its complex with unlabelled barstar have been studied at 40 °C, pH 6.6, using 15N relaxation data obtained from proton-detected 2D {1H}-15N NMR spectroscopy. 15N spin-lattice relaxation rate constants (R1), spin-spin relaxation rate constants (R2), and steady-state heteronuclear {1H}-15N NOEs have been measured at a magnetic field strength of 14.1 Tesla for 91 residues of free barnase and for 90 residues out of a total of 106 in the complex (excluding three prolines and the N-terminal residue) backbone amide 15N sites of barnase. The primary relaxation data for both the cases have been analyzed in the framework of the model-free formalism using both isotropic and axially symmetric models of the rotational diffusion tensor. As per the latter, the overall rotational correlation times (τm) are 5.0 and 9.5 ns for the free and complexed barnase, respectively. The average order parameter is found to be 0.80 for free barnase and 0.86 for the complex. However, the changes are not uniform along the backbone and for about 5 residues near the binding interface there is actually a significant decrease in the order parameters on complex formation. These residues are not involved in the actual binding. For the residues where the order parameter increases, the magnitudes vary significantly. It is observed that the complex has much less internal mobility, compared to free barnase. From the changes in the order parameters, the entropic contribution of NH bond vector motion to the free energy of complex formation has been calculated. It is apparent that these motions cause significant unfavorable contributions and therefore must be compensated by many other favorable contributions to effect tight complex formation. The observed variations in the motion and their different locations with regard to the binding interface may have important implications for remote effects and regulation of the enzyme action.
Similar content being viewed by others
References
Abragam, A. (1961) Principles of Nuclear Magnetism, Clarendon Press, Oxford.
Arcus, V.L., Vuilleumier, S., Freund, S.M., Bycroft, M. and Fersht, A.R. (1995) J. Mol. Biol., 254, 305–321.
Beeser, S.A., Goldenberg, D.P. and Oas, T.G. (1997) J. Mol. Biol., 269, 154–164.
Bhattacharya, S., Falzone, C.J. and Lecomte, T.J. (1999) Biochemistry, 38, 2577–2589.
Bloom, M., Reeves, L.W. and Wells, E.J. (1965) J. Chem. Phys., 42, 1615–1624.
Bycroft, M., Ludvigsen, S., Fersht, A.R. and Poulsen, F.M. (1991) Biochemistry, 30, 8697–8701.
Carr, P.A., Erickson, H.P. and Palmer, A.G., III (1997) Structure, 5, 949–959.
Cavanagh, J., Palmer, A.G., Wright, P.E. and Rance, M. (1991) J.Magn. Reson., 91, 429–436.
Clore, G.M., Szabo, A., Bax, A., Kay, L.E., Driscoll, P.C. and Gronenborn, A.M. (1990a) J. Am. Chem. Soc., 112, 4989–4991.
Clore, G.M., Driscoll, P.C., Wingfield, P.T. and Gronenborn, A.M. (1990b) Biochemistry, 29, 7387–7401.
Epstein, D.M., Benkovic, S.J. and Wright, P.E. (1995) Biochemistry, 34, 11037–11048.
Farrow, N.A., Muhandiram, R., Singer, A.U., Pascal, S.M., Kay, C.M., Gish, G., Shoelson, S.E., Pawson, T., Forman-Kay, J.D. and Kay, L.E. (1994) Biochemistry, 33, 5984–6003.
Fersht, A.R. (1993) FEBS Lett., 325, 5–16.
Fersht, A.R. (1997) Curr. Opin. Struct. Biol., 7, 3–9.
Fischer, M.W.F., Zeng, L., Pang, Y., Hu, W., Majumdar, A. and Zuiderweg, E.R.P. (1997) J. Am. Chem. Soc., 119, 12-629–12-642.
Fitzgerald, P.C. and Hartley, R.W. (1993) Anal. Biochem., 214, 544–547.
Freund, S.M., Wong, K.B. and Fersht, A.R. (1996) Proc. Natl. Acad. Sci. USA, 93, 10600–10603.
Fushman, D., Weisemann, R., Thüring, H. and Rüterjans, H. (1994) J. Biomol. NMR, 4, 61–78.
Fushman, D., Cahill, S. and Cowburn, D. (1997) J. Mol. Biol., 266, 173–194.
Guillet, V., Lapthorn, A., Hartley, R.W. and Mauguen, Y. (1993) Structure, 1, 165–177.
Hiyama, Y., Niu, C.-H., Silverton, J.V., Bavoso, A. and Torchia, D.A. (1988) J. Am. Chem. Soc., 110, 2378–2383.
Hodsdon, M.E. and Cistola, D.P. (1997) Biochemistry, 36, 2278–2290.
Jones, D.N.M., Bycroft, M., Lubienski, M.J. and Fersht, A.R. (1993) FEBS Lett., 331, 165–172.
Kay, L.E., Torchia, D.A. and Bax, A. (1989) Biochemistry, 28, 8972–8979.
Kay, L.E. (1998) Nat. Struct. Biol., 5 (NMR Supplement), 513–517.
Kim, H., Xia, D., Yu, C., Xia, J., Kachurin, A.M., Zhang, L., Yu, L. and Deisenhofer, J. (1998) Proc. Natl. Acad. Sci. USA, 95, 8026–8033.
Khurana, R. and Udgaonkar, J.B. (1994) Biochemistry, 33, 106–115.
Kroenke, C.D., Loria, J.P., Lee, L.K., Rance, M. and Palmer, A.G. II (1998) J. Am. Chem. Soc., 120, 7905–7915.
Lee, A.L., Flynn, P.F. and Wand, A.J. (1999) J. Am. Chem. Soc., 121, 2891–2902.
Lefèvre, J.-F., Dayie, K.T., Peng, J.W. and Wagner, G. (1996) Biochemistry, 35, 2674–2686.
LeMaster, D.M. (1999) J. Am. Chem. Soc., 121, 1726–1742.
Lipari, G. and Szabo, A. (1982a) J. Am. Chem. Soc., 104, 4546–4559.
Lipari, G. and Szabo, A. (1982b) J. Am. Chem. Soc., 104, 4559–4570.
Lubienski, M.J., Bycroft, M., Freund, S.M.V. and Fersht, A.R. (1994) Biochemistry, 33, 8866–8877.
Luginbühl, P., Pervushin, K.V., Iwai, H. and Wüthrich, K. (1997) Biochemistry, 36, 7305–7312.
Malmendal, A., Evenäs, J., Forsen, S. and Akke, M. (1999) J. Mol.Biol., 293, 883–899.
Mandel, A.M., Akke, M. and Palmer, A.G., III (1995) J. Mol. Biol., 246, 144–163.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989) J. Magn.Reson., 85, 393–399.
Mauguen, Y., Hartley, R.W., Dobson, G., Bricogene, G., Chothia, C. and Jack, A. (1982) Nature (London), 297, 162–164.
Matouschek, A., Serrano, L. and Fersht, A.R. (1992) J. Mol. Biol., 224, 819–835.
Mime, S., Tale, S., Ueda, T., Kainosho, M. and Imoto, T. (1999) J.Mol. Biol., 286, 1547–1565.
Okorokov, A.L., Hartley, R.W. and Panov, K.I. (1994) Protein Express. Purif., 5, 547–552.
Palmer, A.G., III, Cavanagh, J., Wright, P.E. and Rance, M. (1991) J. Magn. Reson., 93, 151–170.
Palmer, A.G., III, Williams, J. and McDermott, A. (1996) J. Phys.Chem., 100, 13293–13310.
Peng, J.W. and Wagner, G. (1994) Methods Enzymol., 239, 563–596.
Prompers, J.J., Groenewegen, A., Hilbers, C.W. and Pepermans, H.A.M. (1999) Biochemistry, 38, 5315–5327.
Sanz, J.M., Johnson, C.M. and Fersht, A.R. (1994) Biochemistry, 33, 11189–11199.
Schurr, J.M., Babcock, H.P. and Fujimoto, B.S. (1994) J. Magn.Reson., B105, 211–224.
Skelton, N.J., Palmer, A.G., Akke, M., Kördel, J., Rance, M. and Chazin, W.J. (1993) J. Magn. Reson., B102, 253–264.
Slijper, J., Boelens, R., Davis, A.L., Konings, R.N.H., Marel, G.A. van der, Boom, J.H. van and Kaptein, R. (1997) Biochemistry, 36, 249–254.
Stivers, J.T., Abeygunawardana, C. and Mildvan, A.S. (1996) Biochemistry, 35, 16036–16047.
Tjandra, N., Garrett, D.S., Gronenborn, A.M., Bax, A. and Clore, G.M. (1997) Nature Struct. Biol., 4, 443–449.
Tjandra, N., Feller, S.E., Pastor, R.W. and Bax, A. (1995) J. Am.Chem. Soc., 117, 12562–12566.
Vis, H., Vorgias, C.E., Wilson, K.S., Kaptein, R. and Boelens, R. (1998) J. Biomol. NMR, 11, 265–277.
Werten, S., Wechselberger, R., Boelens, R., Vliet, P.C. van der, and Kaptein, R. (1999) J. Biol. Chem., 274, 3693–3699.
Yang, D. and Kay, L.E. (1996) J. Mol. Biol., 263, 369–382.
Ye, J., Mayer, K.L. and Stone, M.J. (1999) J. Biomol. NMR, 15, 115–124.
Zhang, P., Dayie, K.T. and Wagner, G. (1997) J. Mol. Biol., 272, 443–455.
Zheng, Z., Czaplicki, J. and Jardetzky, O. (1995) Biochemistry, 34, 5212–5223.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sahu, S.C., Bhuyan, A.K., Udgaonkar, J.B. et al. Backbone dynamics of free barnase and its complex with barstar determined by 15N NMR relaxation study. J Biomol NMR 18, 107–118 (2000). https://doi.org/10.1023/A:1008310402933
Issue Date:
DOI: https://doi.org/10.1023/A:1008310402933