Abstract
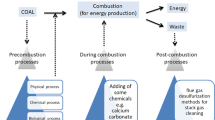
The sulfur found in coal is either part of the molecular coal structure (organically bound sulfur), is contained in minerals such as pyrite (FeS2), or occurs in minor quantities in the form of sulfate and elemental sulfur. When pyrite crystals are finely distributed within the coal matrix, mechanical cleaning can only remove part of the pyrite. It can, however, be removed by microbial action requiring only mild conditions. The process involves simple equipment, almost no chemicals, but relatively long reaction times, and treatment of iron sulfate containing process water. Different process configurations are possibly depending on the coal particle size. Coal with particle sizes of less than 0.5 mm is preferably desulfurised in slurry reactors, while lump coal (> 0.5 mm) should be treated in heaps. Investment and operating costs are estimated for different process configurations on an industrial scale. Concerning the organically bound sulfur in coal there is up to now no promising biochemical pathway for the degradation and/or desulfurisation of such compounds.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klein, J. Technological and economic aspects of coal biodesulfurisation. Biodegradation 9, 293–300 (1998). https://doi.org/10.1023/A:1008282216016
Issue Date:
DOI: https://doi.org/10.1023/A:1008282216016