Abstract
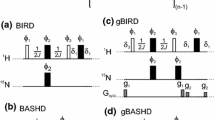
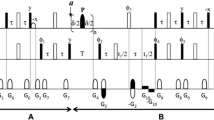
An HMQC experiment is proposed, dubbed FHMQC, where water flip-back is achieved by a single water-selective pulse preceding the basic HMQC pulse sequence. The scheme is demonstrated with a 15N, 1H-HMQC spectrum of uniformly 15N/2H-labelled S. aureus DNA gyrase B with a molecular weight of 45 kDa for the unlabelled protein. The sensitivity of the experiment is improved compared to that of an FHSQC spectrum. It is further shown that the original FHSQC experiment can be shortened by the use of bipolar gradients. Relaxation times of different 15N magnetizations and coherences were measured. The new FHMQC scheme is implemented in 3D NOESY-15N-HMQC and 3D15 N-HMQC-NOESY-15N-HMQC pulse sequences which are demonstrated with a 24 kDa fragment of uniformly 15N/13C/2H-labelled S. aureus DNA gyrase B.
Similar content being viewed by others
References
Bax, A., Griffey, R. H. and Hawkins, B. L. (1983) J. Magn. Reson., 55, 301–315.
Bax, A., Kay, L. E., Sparks, S. W. and Torchia, D. A. (1989) J. Am. Chem. Soc., 111, 408–409.
Bax, A., Ikura, M., Kay, L. E., Torchia, D. A. and Tschudin, R. (1990) J. Magn. Reson., 86, 304–318.
Böckmann, A. and Guittet, E. (1996) J. Biomol. NMR, 8, 87–92.
Bodenhausen, G. and Ruben, D. J. (1980) Chem. Phys. Lett., 69, 185–189.
Boyd, J., Hommel, U. and Campbell, I. D. (1990) Chem. Phys. Lett., 175, 477–482.
Delaglio, F., Grzesiek, S., Vuister, G., Zhu, G., Pfeifer, J. and Bax, A. (1995) J. Biomol. NMR, 6, 277–293.
Farrow, N. A., Muhandiram, R., Singer, A. U., Pascal, S. M., Kay, C. M., Gish, G., Shoelson, S. E., Pawson, T., Forman-Kay, J. D. and Kay, L. E. (1994) Biochemistry, 33, 5984–6003.
Frenkiel, T., Bauer, C., Carr, M. D., Birdsall, B. and Feeney, J. (1990) J. Magn. Reson., 90, 420–425.
Grzesiek, S., Anglister, J., Ren, H. and Bax, A. (1993) J. Am. Chem. Soc., 115, 4369–4370.
Grzesiek, S. and Bax, A. (1993) J. Am. Chem. Soc., 115, 12593–12594.
Hammarström, A. and Otting, G. (1994) J. Magn. Reson., A109, 246–249.
Ikura, M., Bax, A., Clore, G. M. and Gronenborn, A. M. (1990) J. Am. Chem. Soc., 112, 9020–9022.
John, B. K., Plant, D., Webb, P. and Hurd, R. E. (1992) J. Magn. Reson., 98, 200–206.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989a) J. Magn. Reson., 85, 393–399.
Marion, D., Kay, L. E., Sparks, S. W., Torchia, D. A. and Bax, A. (1989b) J. Am. Chem. Soc., 111, 1515–1517.
McCoy, M. A. and Müller, L. (1992) J. Am. Chem. Soc., 114, 2108–2112.
Messerle, B. A., Wider, G., Otting, G., Weber, C. and Wüthrich, K. (1989) J. Magn. Reson., 85, 608–613.
Mori, S., Abeygunawardana, C., Johnson, M. O. and van Zijl, P. C. M. (1995) J. Magn. Reson., B108, 94–98.
Mori, S., Abeygunawardana, C., van Zijl, P. C. M. and Berg, J. M. (1996) J. Magn. Reson., B110, 96–101.
Müller, L. (1979) J. Am. Chem. Soc., 101, 4481–4484.
Nietlispach, D., Clowes, R. T., Broadhurst, R. W., Ito, Y., Keeler, J., Kelly, M., Ashurst, J., Oschkinat, H., Domaille, P. J. and Laue, E. D. (1996) J. Am. Chem. Soc., 118, 407–415.
Norwood, T. J., Boyd, J., Heritage, J. E., Soffe, N. and Campbell, I. D. (1990) J. Magn. Reson., 87, 488–501.
Otting, G. and Liepinsh, E. (1995a) J. Biomol. NMR, 5, 420–426.
Otting, G. and Liepinsh, E. (1995b) J. Magn. Reson., B107, 192–196.
Piotto, M., Saudek, V. and Sklenář, V. (1992) J. Biomol. NMR, 2, 661–665.
Shaka, A. J., Keeler, J., Frenkiel, T. and Freeman, R. (1983) J. Magn. Reson., 52, 335–338.
Shaka, A. J., Barker, P. B. and Freeman, R. (1985) J. Magn. Reson., 64, 547–552.
Shen, J. and Lerner, L. E. (1995) J. Magn. Reson., A112, 265–269.
Skelton, N. J., Palmer III, A. G., Akke, M., Kördel, J., Rance, M. and Chazin, W. J. (1993) J. Magn. Reson., B102, 253–264.
Sklenář, V., Piotto, M., Leppik, R. and Saudek, V. (1993) J. Magn. Reson., A102, 241–245.
Sklenář, V. (1995) J. Magn. Reson., A114, 132–135.
States, D. J., Haberkorn, R. A. and Ruben, D. J. (1982) J. Magn. Reson., 48, 286–292.
Swapna, G. V. T., Rios, C. B., Shang, Z. and Montelione, G. T. (1997) J. Biomol. NMR, 9, 105–111.
Talluri, S. and Wagner, G. (1996) J. Magn. Reson., B112, 200–205.
Van Zijl, P. C. M., Hwang, T. L., Johnson, M. O. and Garwood, M. (1996) J. Am. Chem. Soc., 118, 5510–5511.
Venters, R. A., Huang, C., Farmer, B. T., Trolard, R., Spicer, L. D. and Fierke, C. A. (1995) J. Biomol. NMR, 5, 339–344.
Yamazaki, T., Lee, W., Arrowsmith, C. H., Muhandiram, D. R. and Kay, L. E. (1994) J. Am. Chem. Soc., 116, 11655–11666.
Zhang, O. W., Kay, L. E., Olivier, J. P. and Forman-Kay, J. D. (1994) J. Biomol. NMR, 4, 845–858.
Zuiderweg, E. R. P. and Fesik, S. W. (1989) Biochemistry, 28, 2387–2391.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Andersson, P., Gsell, B., Wipf, B. et al. HMQC and HSQC experiments with water flip-back optimized for large proteins. J Biomol NMR 11, 279–288 (1998). https://doi.org/10.1023/A:1008227631084
Issue Date:
DOI: https://doi.org/10.1023/A:1008227631084