Abstract
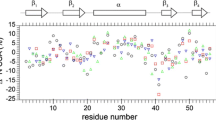
Backbone and tryptophan side-chain mobilities in the 26-residue, cytolytic peptide melittin (MLT) were investigated by 15N and 13C NMR. Specifically, inverse-detected 15N T1 and steady-state NOE measurements were made at 30 and 51 MHz on MLT at 22 °C enriched with 15N at six amide positions and in the Trp19 side chain. Both the disordered MLT monomer (1.2 mM peptide at pH 3.6 in neat water) and α-helical MLT tetramer (4.0 mM peptide at pH 5.2 in 150 mM phosphate buffer) were examined. The relaxation data were analyzed in terms of the Lipari and Szabo model-free formalism with three parameters: τm, the correlation time for the overall rotation; S2, a site-specific order parameter which is a measure of the amplitude of the internal motion; and τe, a local, effective correlation time of the internal motion. A comparison was made of motional parameters from the 15N measurements and from 13C measurements on MLT, the latter having been made here and previously [Kemple et al. (1997) Biochemistry, 36, 1678–1688]. τm and τe values were consistent from data on the two nuclei. In the MLT monomer, S2 values for the backbone N-H and Cα-H vectors in the same residue were similar in value but in the tetramer the N-H order parameters were about 0.2 units larger than the Cα-H order parameters. The Trp side-chain N-H and C-H order parameters, and τe values were generally similar in both the monomer and tetramer. Implications of these results regarding the dynamics of MLT are examined.
Similar content being viewed by others
References
Barbato, G., Ikura, M., Kay, L.E., Pastor, R.W. and Bax, A. (1992) Biochemistry, 31, 5269–5278.
Brown, L.R, Lauterwein, J. and Wüthrich, K. (1980) Biochim. Biophys. Acta, 622, 231–244.
Buckley, P., Edison, A.S., Kemple, M.D. and Prendergast, F.G. (1993) J. Biomol. NMR, 3, 639–652.
Chandrasekhar, I., Clore, G.M., Szabo, A., Gronenborn, A.M. and Brooks, B.R. (1992) J. Mol. Biol., 226, 239–250.
Clore, G.M., Szabo, A., Bax, A., Kay, L.E., Driscoll, P.C. and Gronenborn, A.M. (1990) J. Am. Chem. Soc., 112, 4989–4991.
Cross, T.A. and Opella, S.J. (1983) J. Am. Chem. Soc., 105, 306–308.
Daggett, V., Kollman, P.A. and Kuntz, I.D. (1991) Biopolymers, 31, 1115–1134.
Dempsey, C.E. (1990) Biochim. Biophys. Acta, 1031, 143–161.
Eriksson, M.A.L., Berglund, H., Härd, T. and Nilsson, L. (1993) Proteins Struct. Funct. Genet., 17, 375–390.
Fadel, A.R., Jin, D.Q., Montelione, G.T. and Levy, R.M. (1995) J. Biomol. NMR, 6, 221–226.
Farrow, N.A., Zhang, O., Szabo, A., Torchia, D.A. and Kay, L.E. (1995) J. Biomol. NMR, 6, 153–162.
Fox, T. and Kollman, P.A. (1996) Proteins Struct. Funct. Genet., 25, 315–334.
Fushman, D., Cahill, S. and Cowburn, D. (1997) J. Mol. Biol., 266, 173–194.
Grzesiek, R. and Bax, A. (1993) J. Am. Chem. Soc., 115, 12593–12594.
Hiyama, Y., Niu, C., Silverton, J.V., Bavoso, A. and Torchia, D.A. (1988) J. Am. Chem. Soc., 110, 2378–2383.
Ishima, R., Yamasaki, K. and Nagayama, K. (1995a) J. Biomol. NMR, 6, 423–426.
Ishima, R., Yamasaki, K., Saito, M. and Nagayama, K. (1995b) J. Biomol. NMR, 6, 217–220.
Kay, L.E., Torchia, D.A. and Bax, A. (1989) Biochemistry, 28, 8972–8979.
Kemple, M.D., Yuan, P., Nollet, K.E., Fuchs, J.A., Silva, N. and Prendergast, F.G. (1994) Biophys. J., 66, 2111–2126.
Kemple, M.D., Buckley, P., Yuan, P. and Prendergast, F.G. (1997) Biochemistry, 36, 1678–1688.
Kinosita, K.S., Kawato, S. and Ikegami, A. (1977) Biophys. J., 20, 289–305.
Kördel, J., Skelton, N.J., Akke, M., Palmer, A.G. and Chazin, W.J. (1992) Biochemistry, 31, 4856–4866.
Kördel, J. and Teleman, O. (1992) J. Am. Chem. Soc., 114, 4934–4936.
Lauterwein, J., Brown, L.R. and Wüthrich, K. (1980) Biochim. Biophys. Acta, 622, 219–230.
Lee, L.K., Rance, M., Chazin, W.J. and Palmer, A.G. (1997) J. Biomol. NMR, 9, 287–298.
Lefèvre, J.-F., Dayie, K.T., Peng, J.W. and Wagner, G. (1996) Biochemistry, 35, 2674–2686.
LeMaster, D.M. and Kushlan, D.M. (1996) J. Am. Chem. Soc., 118, 9255–9264.
Lipari, G. and Szabo, A. (1982) J. Am. Chem. Soc., 104, 4546–4559.
London, R.E. and Avitabile, J. (1978) J. Am. Chem. Soc., 100, 7159–7165.
Mandel, A.M., Akke, M. and Palmer, A.G. (1995) J. Mol. Biol., 246, 144–163.
Palmer, A.G. and Case, D.A. (1992) J. Am. Chem. Soc., 114, 9059–9067.
Palmer, A.G., Hochstrasser, R.A., Millar, D.P., Rance, M. and Wright, P.E. (1993) J. Am. Chem. Soc., 115, 6333–6345.
Peng, J.W. and Wagner, G. (1995) Biochemistry, 34, 16733–16752.
Schneider, D.M., Dellow, M.J. and Wand, A.J. (1992) Biochemistry, 31, 3645–3652.
Shaka, A.J., Keeler, J., Frenkiel, T. and Freeman, R. (1983) J. Magn. Reson., 52, 335–338.
Shaka, A.J., Barker, P.B. and Freeman, R. (1985) J. Magn. Reson., 64, 547–552.
Smith, L.J., Mark, A.E., Dobson, C.M. and van Gunsteren, W.F. (1995a) Biochemistry, 34, 10918–10931.
Smith, P.E., Schaik, R.C., Szyperski, T., Wüthrich, K. and van Gunsteren, W.F. (1995b) J. Mol. Biol., 246, 356–365.
Stone, M.J., Fairbrother, W.J., Palmer, A.G., Reizer, J., Saier, M.H. and Wright, P.E. (1992) Biochemistry, 31, 4394–4406.
Stone, M.J., Chandrasekhar, K., Holmgren, A., Wright, P.E. and Dyson, H.J. (1993) Biochemistry, 32, 426–435.
Terwilliger, T.C. and Eisenberg, D. (1982a) J. Biol. Chem., 257, 6010–6015.
Terwilliger, T.C. and Eisenberg, D. (1982b) J. Biol. Chem., 257, 6016–6022.
Torchia, D.A., Nicholson, L.K., Cole, H.B.R. and Kay, L.E. (1993) In NMR of Proteins (Eds., Clore, G.M. and Gronenborn, A.M.), CRC Press, Boca Raton, FL, pp. 190–219.
Wand, A.J., Urbauer, J.L., McEvoy, R.P. and Bieber, R.J. (1996) Biochemistry, 35, 6116–6125.
Weaver, A.J., Kemple, M.D. and Prendergast, F.G. (1989) Biochemistry, 28, 8624–8639.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991) J. Mol. Biol., 222, 311–333.
Wishart, D.S., Bigam, C.G., Yao, J., Abildgaard, F., Dyson, H.J., Oldfield, E., Markley, J.L. and Sykes, B.D. (1995) J. Biomol. NMR, 6, 135–140.
Yamasaki, K., Saito, M., Oobatake, M. and Kanaya, S. (1995) Biochemistry, 34, 6587–6601.
Yamazaki, T., Muhandiram, R. and Kay, L.E. (1994) J. Am. Chem. Soc., 116, 8266–8278.
Yuan, P., Fisher, P.J., Prendergast, F.G. and Kemple, M.D. (1996) Biophys. J., 70, 2223–2238.
Zheng, Z., Czaplicki, J. and Jardetzky, O. (1995) Biochemistry, 34, 5212–5223.
Zhu, L., Kemple, M.D., Yuan, P. and Prendergast, F.G. (1995) Biochemistry, 34, 13196–13202.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhu, L., Prendergast, F.G. & Kemple, M.D. Comparison of 15N- and 13C-determined parameters of mobility in melittin. J Biomol NMR 12, 135–144 (1998). https://doi.org/10.1023/A:1008210613711
Issue Date:
DOI: https://doi.org/10.1023/A:1008210613711