Abstract
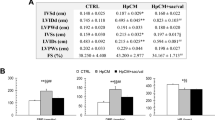
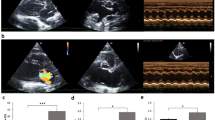
The purpose of this study is to determine whether the administration of the ACE inhibitor cilazapril can lessen the adverse effects of ventricular remodeling, including systolic and diastolic dysfunction, modulation of fetal gene expression, increase of collagen genes, and depression of the sarcoplasmic reticulum (SR) Ca2+ ATPase gene in a myocardial infarcted (MI) rat model. At 1 day after MI, the animals were randomly assigned to cilazapril treatment or no treatment. We performed Doppler-echocardiographic examinations and measured cardiac mRNA in rats at 1 month and 3 months after MI (each group n = 8). The weights of the right (RV) and left ventricles (LV) in 1- and 3-month MI rats were significantly larger than those of the control rats. Cilazapril significantly prevented the increase. The MI rats showed systolic dysfunction, as evidenced by decreased fractional shortening (control, 34 ± 3% vs. MI, 17 ± 3%; P <0.01) and ejection fraction measured by the modified Simpson’s method (control, 61 ± 2% vs. MI, 36 ± 3%; P < 0.01) in rats at 1 month after operation. MI rats showed diastolic dysfunction, defined as increased peak early filling velocity, increased deceleration rate of the early filling wave, decreased late filling velocity, and an increase in the ratio of early filling to late filling velocity. Cilazapril significantly prevented systolic and diastolic dysfunction in rats after MI. The increases in β-MHC, α-skeletal actin, ANP, and collagen I and III mRNAs in the nonischemic LV and RV were significantly suppressed by treatment with cilazapril. Depressed SR Ca2+-ATPase mRNA (nonischemic LV, 0.7-fold, P < 0.05 vs. control; RV, 0.5-fold, P < 0.05 vs. control) at 3 months after MI was significantly restored to normal levels by cilazapril. Cilazapril improved the adverse remodeling process by attenuating the progression of systolic and diastolic dysfunction, and prevented abnormal cardiac gene expression following MI.
Similar content being viewed by others
References
Pfeffer MA, Braunwald E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990;81:1161-1172.
Weisman HF, Bush DE, Mannisi JA, Bulkley BH. Global cardiac remodeling after acute myocardial infarction: A study in the rat model. J Am Coll Cardiol 1985;5:1355-1362.
McKay RG, Pfeffer MA, Pasternak RC, et al. Left ventricular remodeling after myocardial infarction: A corollary to infarct expansion. Circulation 1986;74:693-702.
Anversa P, Loud AV, Levicky V, Guideri G. Left ventricular failure induced by myocardial infarction. I. Myocyte hypertrophy. Am J Physiol 1985;248:H883-H889.
Fishbein MC, Maclean D, Maroko PR. Experimental myocardial infarction in the rat: Qualitative and quantitative changes during pathologic evolution. Am J Pathol 1978;90: 57-70.
Sun Y, Weber KT. Angiotensin II receptor binding following myocardial infarction in the rat. Cardiovasc Res 1994;28: 1623-1628.
Sun Y, Cleutjens JP, Diaz-Arias AA, Weber KT. Cardiac angiotensin converting enzyme and myocardial fibrosis in the rat [see comments]. Cardiovasc Res 1994;28:1423-1432.
Sun Y, Weber KT. Angiotensin converting enzyme and myofibroblasts during tissue repair in the rat heart. J Mol Cell Cardiol 1996;28:851-858.
Sun Y, Weber KT. Cells expressing angiotensin II receptors in fibrous tissue of rat heart. Cardiovasc Res 1996;31: 518-525.
CONSENSUS. Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987;316:1429-1435.
SOLVD. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators [see comments]. N Engl J Med 1991;325:293-302.
Pfeffer MA, Pfeffer JM, Steinberg C, Finn P. Survival after an experimental myocardial infarction: Beneficial effects of long-term therapy with captopril. Circulation 1985;72: 406-412.
Pfeffer MA, Braunwald E, Moye LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the Survival And Ventricular Enlargement trial. The SAVE Investigators [see comments]. N Engl J Med 1992;327: 669-677.
Passier RC, Smits JF, Verluyten MJ, Daemen MJ. Expression and localization of renin and angiotensinogen in rat heart after myocardial infarction. Am J Physiol 1996;271: H1040-H1048.
Schunkert H, Tang SS, Litwin SE, Diamant D, Riegger G, Dzau VJ, Ingelfinger JR. Regulation of intrarenal and circulating renin-angiotensin systems in severe heart failure in the rat. Cardiovasc Res 1993;27:731-735.
Dzau VJ. Implications of local angiotensin production in cardiovascular physiology and pharmacology. Am J Cardiol 1987;59:59A-65A.
Hirsch AT, Talsness CE, Schunkert H, Paul M, Dzau VJ. Tissue-specific activation of cardiac angiotensin converting enzyme in experimental heart failure. Circ Res 1991;69: 475-482.
Hodsman GP, Kohzuki M, Howes LG, Sumithran E, Tsunoda K, Johnston CI. Neurohumoral responses to chronic myocardial infarction in rats. Circulation 1988;78:376-381.
Lindpaintner K, Ganten D. The cardiac renin-angiotensin system. An appraisal of present experimental and clinical evidence. Circ Res 1991;68:905-921.
Yamagishi H, Kim S, Nishikimi T, Takeuchi K, Takeda T. Contribution of cardiac renin-angiotensin system to ventricular remodelling in myocardial-infarcted rats. J Mol Cell Cardiol 1993;25:1369-1380.
Gay RG. Early and late effects of captopril treatment after large myocardial infarction in rats. J Am Coll Cardiol 1990;16:967-977.
Kim S, Ohta K, Hamaguchi A, Yukimura T, Miura K, Iwao H. Angiotensin II induces cardiac phenotypic modulation and remodeling in vivo in rats. Hypertension 1995;25: 1252-1259.
Genovese C, Rowe D, Kream B. Construction of DNA sequences complementary to rat alpha 1 and alpha 2 collagen mRNA and their use in studying the regulation of type I collagen synthesis by 1,25-dihydroxyvitamin D. Biochemistry 1984;23:6210-6216.
Liau G, Yamada Y, de CB. Coordinate regulation of the levels of type III and type I collagen mRNA in most but not all mouse fibroblasts. J Biol Chem 1985;260:531-536.
MacLennan DH, Brandl CJ, Korczak B, Green NM. Aminoacid sequence of a Ca2+ + Mg2+-dependent ATPase from rabbit muscle sarcoplasmic reticulum, deduced fromits complementary DNA sequence. Nature 1985;316:696-700.
Kim S, Ohta K, Hamaguchi A, et al. Role of angiotensin II in renal injury of deoxycorticosterone acetate-salt hypertensive rats. Hypertension 1994;24:195-204.
Xie GY, Berk MR, Smith MD, Gurley JC, DeMaria AN. Prognostic value of Doppler transmitral flow patterns in patients with congestive heart failure [see comments]. J Am Coll Cardiol 1994;24:132-139.
Giannuzzi P, Imparato A, Temporelli PL, de-Vito F, Silva PL, Scapellato F, Giordano A. Doppler-derived mitral deceleration time of early filling as a strong predictor of pulmonary capillary wedge pressure in postinfarction patients with left ventricular systolic dysfunction. J Am Coll Cardiol 1994;23:1630-1637.
Hanatani A, Yoshiyama M, Kim S, et al. Inhibition by angiotensin II type 1 receptor antagonist of cardiac phenotypic modulation after myocardial infarction. J Mol Cell Cardiol 1995;27:1905-1914.
Chassagne C, Wisnewsky C, Schwartz K. Antithetical accumulation of myosin heavy chain but not alpha-actin mRNA isoforms during early stages of pressure-overload-induced rat cardiac hypertrophy. Circ Res 1993;72:857-864.
Schiaffino S, Samuel JL, Sassoon D, et al. Nonsynchronous accumulation of alpha-skeletal actin and beta-myosin heavy chain mRNAs during early stages of pressure-overload-induced cardiac hypertrophy demonstrated by in situ hybridization. Circ Res 1989;64:937-948.
Swynghedauw B. Developmental and functional adaptation of contractile proteins in cardiac and skeletal muscles. Physiol Rev 1986;66:710-771.
Hewett TE, Grupp IL, Grupp G, Robbins J. Alpha-skeletal actin is associated with increased contractility in the mouse heart. Circ Res 1994;74:740-746.
Izumo S, Nadal GB, Mahdavi V. Protooncogene induction and reprogramming of cardiac gene expression produced by pressure overload. Proc Natl Acad Sci USA 1988;85: 339-343.
Bers DM, Bridge JH. Relaxation of rabbit ventricular muscle by Na-Ca exchange and sarcoplasmic reticulum calcium pump. Ryanodine and voltage sensitivity. Circ Res 1989;65: 334-342.
Bers DM, Lederer WJ, Berlin JR. Intracellular Ca transients in rat cardiac myocytes: Role of Na-Ca exchange in excitation-contraction coupling. Am J Physiol 1990;258: C944-C954.
Katz AM. Basic cellular mechanisms of action of the calcium-channel blockers. Am J Cardiol 1985;55:2B-9B.
Bentivegna LA, Ablin LW, Kihara Y, Morgan JP. Altered calcium handling in left ventricular pressure-overload hypertrophy as detected with aequorin in the isolated, perfused ferret heart. Circ Res 1991;69:1538-1545.
Arai M, Alpert NR, MacLennan DH, Barton P, Periasamy M. Alterations in sarcoplasmic reticulum gene expression in human heart failure. A possible mechanism for alterations in systolic and diastolic properties of the failing myocardium. Circ Res 1993;72:463-469.
Volders PG, Willems IE, Cleutjens JP, Arends JW, Havenith MG, Daemen MJ. Interstitial collagen is increased in the non-infarcted human myocardium after myocardial infarction. J Mol Cell Cardiol 1993;25:1317-1323.
Weber KT, Sun Y, Tyagi SC, Cleutjens JP. Collagen network of the myocardium: Function, structural remodeling and regulatory mechanisms. J Mol Cell Cardiol 1994;26:279-292.
Weber KT, Janicki JS, Shroff SG, Pick R, Chen RM, Bashey RI. Collagen remodeling of the pressure-overloaded, hypertrophied nonhuman primate myocardium. Circ Res 1988;62:757-765.
Weber KT, Sun Y, Campbell SE. Structural remodelling of the heart by fibrous tissue: Role of circulating hormones and locally produced peptides. Eur Heart J 1995;16(Suppl. N): 12-18.
Boluyt MO, O'Neill L, Meredith AL, et al. Alterations in cardiac gene expression during the transition from stable hypertrophy to heart failure. Marked upregulation of genes encoding extracellular matrix components. Circ Res 1994;75:23-32.
Weber KT, Brilla CG. Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation 1991;83:1849-1865.
van-Krimpen C, Schoemaker RG, Cleutjens JP, Smits JF, Struyker-Boudier HA, Bosman FT, Daemen MJ. Angiotensin I converting enzyme inhibitors and cardiac remodeling. Basic Res Cardiol 1991;1:149-155.
van-Krimpen C, Smits JF, Cleutjens JP, et al. DNA synthesis in the non-infarcted cardiac interstitium after left coronary artery ligation in the rat: Effects of captopril. J Mol Cell Cardiol 1991;23:1245-1253.
Smits JF, van KC, Schoemaker RG, Cleutjens JP, Daemen MJ. Angiotensin II receptor blockade after myocardial infarction in rats: Effects on hemodynamics, myocardial DNA synthesis, and interstitial collagen content. J Cardiovasc Pharmacol 1992;20:772-778.
Meggs LG, Coupet J, Huang H, et al. Regulation of angiotensin II receptors on ventricular myocytes after myocardial infarction in rats. Circ Res 1993;72:1149-1162.
Ichikawa I, Pfeffer JM, Pfeffer MA, Hostetter TH, Brenner BM. Role of angiotensin II in the altered renal function of congestive heart failure. Circ Res 1984;55:669-675.
Raya TE, Gay RG, Aguirre M, Goldman S. Importance of venodilatation in prevention of left ventricular dilatation after chronic large myocardial infarction in rats: A comparison of captopril and hydralazine. Circ Res 1989;64:330-337.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshiyama, M., Takeuchi, K., Hanatani, A. et al. Effect of Cilazapril on Ventricular Remodeling Assessed by Doppler-Echocardiographic Assessment and Cardiac Gene Expression. Cardiovasc Drugs Ther 12, 57–70 (1998). https://doi.org/10.1023/A:1007789519005
Issue Date:
DOI: https://doi.org/10.1023/A:1007789519005