Abstract
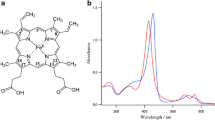
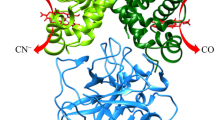
The interaction of apohemoglobin with two heme derivatives, CN-protohemin and CN-deuterohemin, was monitored at multiple Soret wavelengths (417–423 and 406–412 nm, respectively) in 0.05 M potassium phosphate buffer, pH 7.0, at 10°C and revealed, as previously reported, a multiphasic kinetic reaction. Wavelength-dependent reactions were observed for both CN-protohemin and CN-deuterohemin derivatives with the a chain (bathochromic entity) displaying faster (4- to 7-fold) rates throughout the courses of both heme-binding reactions. The basis of this spectrally heterogeneous kinetic phenomenon could be deduced from molecular modeling studies of α- and β-chain structures. Key differences in the number of stabilizing contacts of the two chains with the peripheral a propionyl 45(CE3); 58(E7); 61(E10) as well as the b vinyl 38(C4); 71(E15); 106(G8) groups were found. Furthermore, RMS plots comparing apo- and heme-containing subunits reveal substantial structural disparities in the C-CD-F-FG helical regions of the αβ dimer interface.
Similar content being viewed by others
REFERENCES
Aojula, H. S., Wilson, M. T., and Drake, A. (1986). Biochem. J. 237, 613–616.
Baldwin, J. M. (1980). J. Mol. Biol. 136, 103–128.
Cassoly, R., Bucci, E., Iwatsubo, M., and Bannerjee, R. (1967). Biochem. Biophys. Acta 133, 557–567.
Chiu, F., Vasudevan, G., Morris, A., and McDonald, M. J. (1998). Biochem. Biophys. Res. Commun. 242, 365–368.
Craik, C. S., Buchman, S. R., and Beychok, S. (1980). Proc. Natl. Acad. Sci. USA 77, 1384–1388.
Creighton, T. E. (1993). Proteins. Structures and Molecular Properties, Freeman; New York.
Fermi, G. and Perutz, M. F. (1981). Haemoglobin and Myoglobin. Atlas of Molecular Structures in Biology (Philips, D. C., and Richards, F. M., eds.), Clarendon Press, Oxford.
Guex, N., Diemand, A., and Pietsch, M. C. (1999). Trends Biol. Sci. 24, 364–367.
Gibson, Q. H. and Antonini, E. (1960). J. Biochem. 77, 328–341.
Gibson, Q. H. and Antonini, E. (1963). J. Biol. Chem. 238, 1384–1388.
Inaba, K., Ishimori, K., Imai, K., and Morishima, I. (2000). J. Biol. Chem. 275, 12438–12445.
Ishimori, K. and Morishima, I. (1988). Biochemistry 27, 4747–4753.
Kawamura-Konishi, Y. and Suzuki, H. (1985). J. Biochem. 98, 1181–1190.
Kawamura-Konishi, Y., Chiba, K., Kihara, H., and Suzuki, H. (1992). Eur. Biophys. J. 21, 85–92.
Kolczak, U., Han, C., Sylvia, L. A., and La Mar, G. N. (1997). J. Am. Chem. Soc. 119, 12643–12654.
Kolczak, U., Hauksson, J. B., Davis, N. L., Pande, U., de Ropp, J. S., Langry, K. C., Smith, K. M., and La Mar, G. N. (1999). J. Am. Chem. Soc. 121, 835–843.
La Mar, G. N., Yamamoto, Y., Jue, T., Smith, K. M., and Pandey, R. K. (1985). Biochemistry 24, 3826–3831.
Moulton, D. P. and McDonald, M. J. (1994). Biochem. Biophys. Res. Commun. 199, 1278–1283.
Park, R. Y. and McDonald, M. J. (1989). Biochem. Biophys. Res. Commun. 162, 522–527.
Peitsch, M. C. (1996). Biochem. Soc. Trans. 24, 274–279.
Rose, M. Y. and Olson, J. S. (1983). J. Biol. Chem. 258, 4298–4303.
Schaad, O., Vallone, B., and Edelstein, S. J. (1993). C. R. Acad. Sci. Paris 316, 564–571.
Sobolev, V., Sorokine, A., Prilusky, J., Abola, E. E., and Edelmen, M. (1999). Bioinformatics 15, 327–332.
Vasudevan, G. and McDonald, M. J. (1997). J. Biol. Chem. 272, 517–524.
Vasudevan, G. and McDonald, M. J. (1998). J. Protein Chem. 17, 319–327.
Winterhalter, K. H. and Glathaar, B. (1971). Ser. Haemat. 4, 84–96.
Winterhalter, K. H., Ioppolo, C., and Antonini, E. (1971). Biochemistry 10, 3790–3795.
Yip, Y. K., Waks, M., and Beychok, S. (1977). Proc. Natl. Acad. Sci. USA 74, 64–68.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasudevan, G., McDonald, M.J. Wavelength-Dependent Spectral Changes Accompany CN-Hemin Binding to Human Apohemoglobin. J Protein Chem 19, 583–590 (2000). https://doi.org/10.1023/A:1007150318854
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1007150318854